Research Article
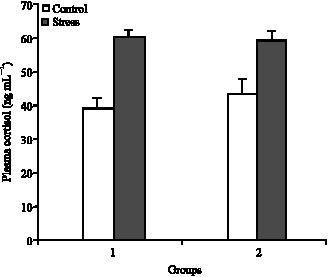
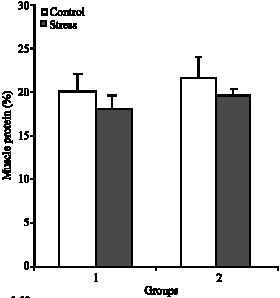
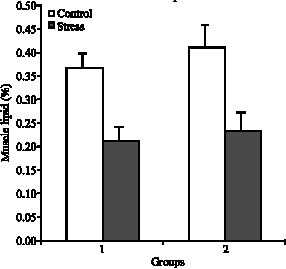
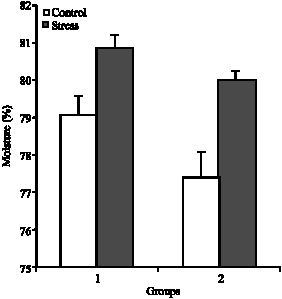
Plasma Cortisol Changes and Body Composition in Stizostedion lucioperca Exposed to Handling Stress
Department of Biology, University of Alzahra, Tehran, Iran
Ghafori Sanaz
Department of Marine Biology, Islamic Azad University, Tehran, Iran
Jamili Shahla
Department of Marine Biology, Islamic Azad University, Tehran, Iran