Research Article
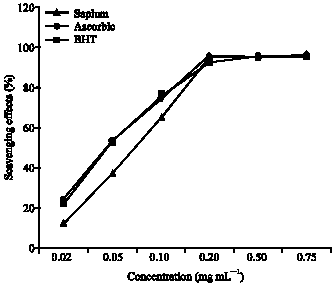
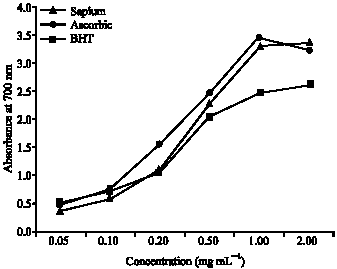
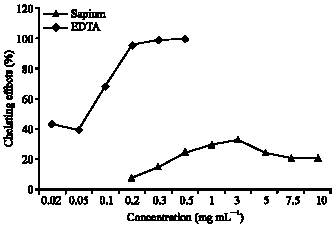
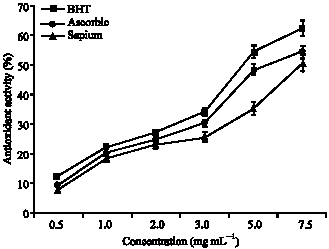
Antioxidant Activities of Methanolic Extract of Sapium ellipticum
Department of Pharmacognosy, University of Lagos, P.M.B. 12003, Lagos, Nigeria
N.A. Elechi
Department of Pharmacognosy, University of Lagos, P.M.B. 12003, Lagos, Nigeria
H.A.B. Coker
Department of Pharmaceutical Chemistry, Faculty of Pharmacy, University of Lagos, P.M.B. 12003, Lagos, Nigeria