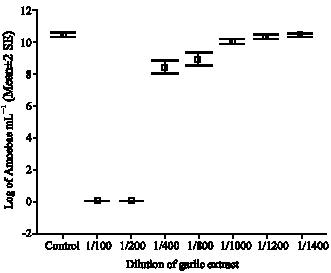
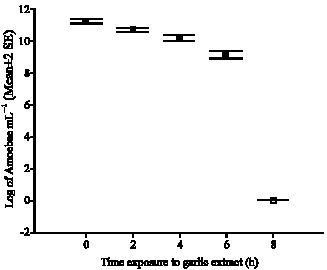
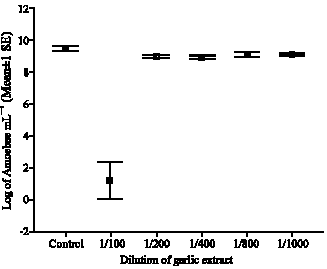
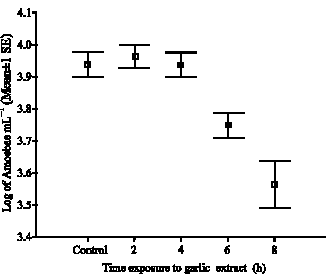
Research Article
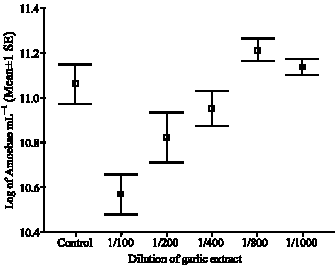
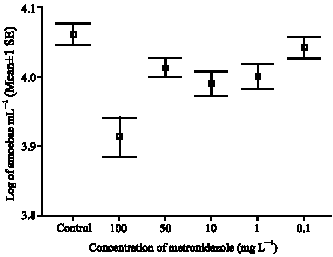
In vitro Effect of Garlic Extract and Metronidazole Against Neoparamoeba pemaquidensis, Page 1987 and Isolated Amoebae from Atlantic Salmon
Faculty of Veterinary, Shahid Chamran University of Ahvaz, P.O. Box 145, Ahvaz, Iran
M.D. Powell
School of Aquaculture, Tasmanian Aquaculture and FisheriesInstitute, The Cooperative Research Centre for Sustainable Development of Fin Fish Aquaculture, University of Tasmania, Locked Bag 1370, Launceston, Tasmania, 7250, Australia
M.R. Zadkarami
Department of Statistics, Faculty of Mathematics,Shahid Chamran University, Ahvaz, Iran