Research Article
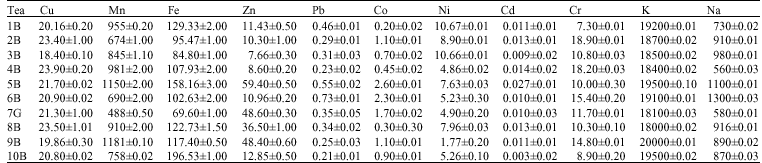
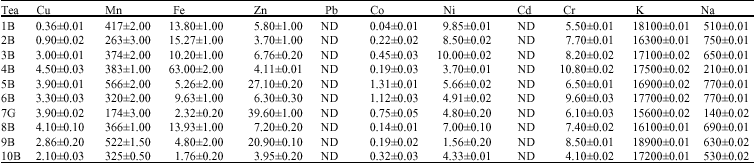
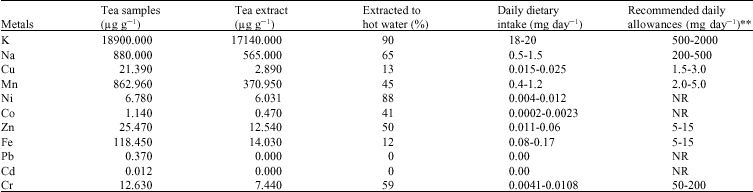
Quantitative Assessment of Metals in Local Brands of Tea in Pakistan
International Center for Chemical and Biological Sciences, HEJ Research Institute of Chemistry, University of Karachi, Pakistan
E. Zahir
Department of Chemistry, University of Karachi, 75270, Karachi, Pakistan
S. Mohiuddin
Department of Chemistry, University of Karachi, 75270, Karachi, Pakistan
A. Nisar Khan
International Center for Chemical and Biological Sciences, HEJ Research Institute of Chemistry, University of Karachi, Pakistan
I.I. Naqvi
Department of Chemistry, University of Karachi, 75270, Karachi, Pakistan