Research Article
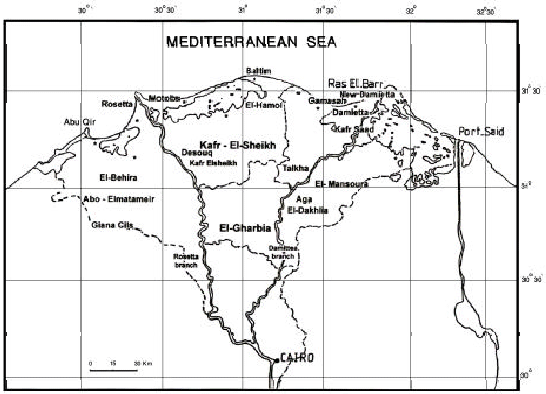
Habitats and Plant Communities in the Nile Delta of Egypt I. Deltaic Mediterranean Coastal Habitat
Department of Botany, Faculty of Science, Mansoura University, Mansoura, Egypt
I.E. El-Habashy
Department of Botany, Faculty of Science, Mansoura University, Mansoura, Egypt
E.F. El-Halawany
Department of Botany, Faculty of Science, Mansoura University, Mansoura, Egypt
G. Omar
Department of Botany, Faculty of Science, Mansoura University, Mansoura, Egypt