Research Article
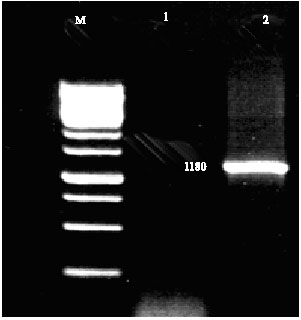
Expression of Bovine leukemia virus p24 Protein in Bacterial Cell
Department of Microbiology, Faculty of Veterinary Medicine, Islamic Azad University of Shahrekord, Shahrekord, Iran
F. Hemmatzadeh
Department of Microbiology, Faculty of Veterinary Medicine, University of Tehran, P.O. Box 1166, Tehran, Iran
H. Keyvanfar
Department of Microbiology, Faculty of Veterinary Medicine, University of Tehran, P.O. Box 1166, Tehran, Iran