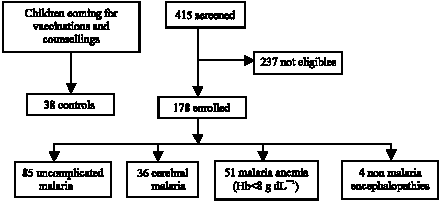
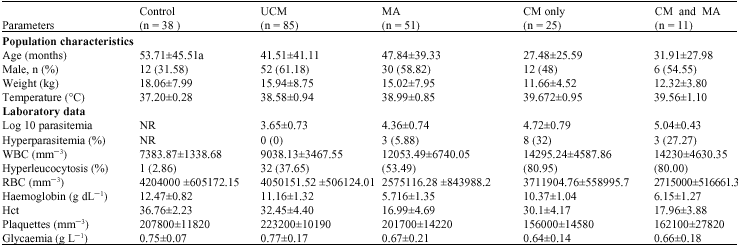
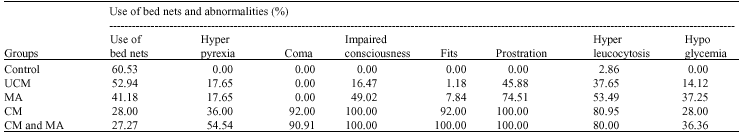
Research Article
Clinical Presentation, Haematological Indices and Management of Children with Severe and Uncomplicated Malaria in Douala, Cameroon
Department of Biochemistry, Faculty of Science, University of Douala, Cameroon
I. Gouado
Department of Biochemistry, Faculty of Science, University of Douala, Cameroon
H. Fotso Kuate
Laboratory Service, Laquintinie Hospital, Douala, Cameroon
O. Zambou
Paediatric Service, Deido District Hospital, Douala, Cameroon
G. Grau
Vascular Immuno-Pathology Unit, Faculty of Medicine, University of Sydney, Australia
V. Combes
Vascular Immuno-Pathology Unit, Faculty of Medicine, University of Sydney, Australia
P.H. Amvam Zollo
Department of Biochemistry, Faculty of Science, University of Douala, Cameroon