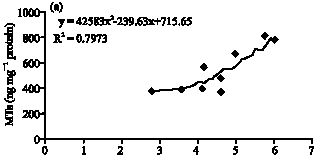
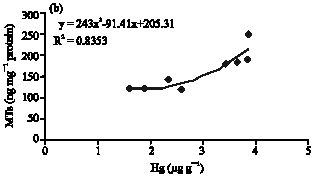
Clinical Report
Evaluation of Metallothionein Expression as a Biomarker of Mercury Exposure in Scatophagus argus
Khoramshahr University of Marine Science and Technology, Iran
H. Zolgharnein
Khoramshahr University of Marine Science and Technology, Iran
M. Sinaei
Khoramshahr University of Marine Science and Technology, Iran
H. Najafzadeh
Chamran University, Ahvaz, Iran
M. Ghavasi
Khoramshahr University of Marine Science and Technology, Iran