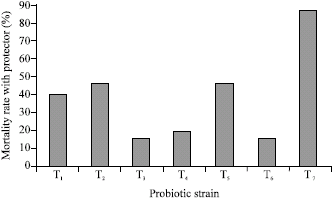
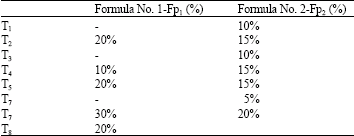
Research Article
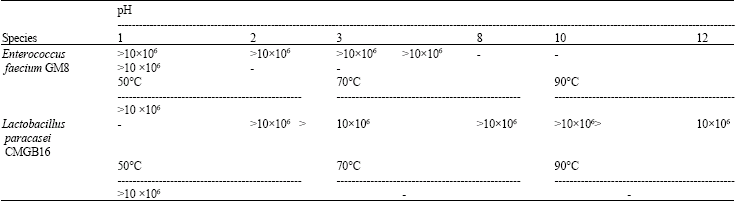
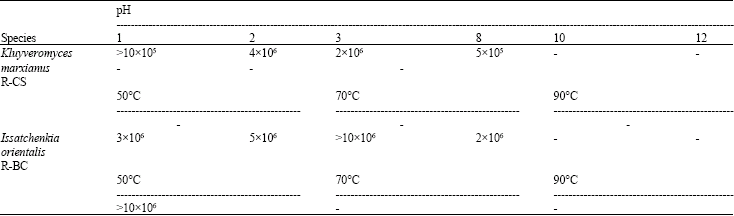
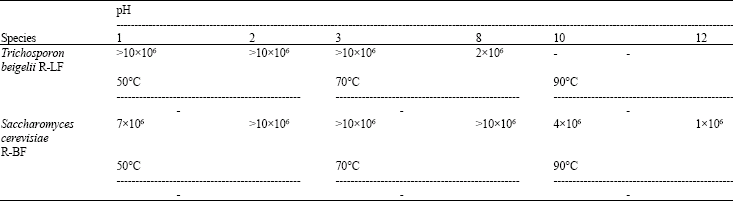
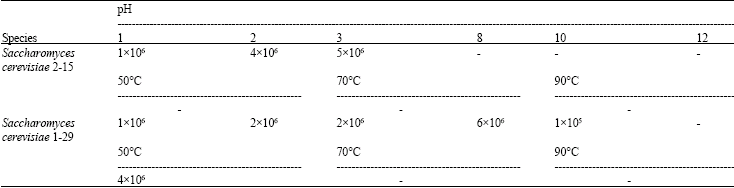
Effect of the Yeast and Bacteria Biomass on the Microbiota in the Rumen
Faculty of Biotechnology, University of Agronomic Sciences and Veterinary Medicine, Bd. Marasti No. 59, District 1, Bucharest, Romania
A. Vamanu
Faculty of Biotechnology, University of Agronomic Sciences and Veterinary Medicine, Bd. Marasti No. 59, District 1, Bucharest, Romania
O. Popa
Faculty of Biotechnology, University of Agronomic Sciences and Veterinary Medicine, Bd. Marasti No. 59, District 1, Bucharest, Romania
Tatiana Vassu
Faculty of Biology, University of Bucharest, Splaiul Independenþei No. 91-95, District 5, Zip 76201, Bucharest, Romania
Raluca Ghindea
Faculty of Biology, University of Bucharest, Splaiul Independenþei No. 91-95, District 5, Zip 76201, Bucharest, Romania
Diana Pelinescu
Faculty of Biology, University of Bucharest, Splaiul Independenþei No. 91-95, District 5, Zip 76201, Bucharest, Romania
Sultana Nita
National Institute of Chemical-Pharmaceutical Research-Development-ICCF Bucharest, Vitan Road No. 112, sector 3, Bucharest, Romania
Narcisa Babeanu
Faculty of Biotechnology, University of Agronomic Sciences and Veterinary Medicine, Bd. Marasti No. 59, District 1, Bucharest, Romania