ABSTRACT
The study has been carried out under the greenhouse and laboratory conditions, using the lyophilization technique to determine the preserving possibilities of 3 native strain isolated from bean root nodules that been widely cultivated in Turkey and was defined as Rhizobium bacteria. The strains been developed separately in the Petri dishes with YMA (Yeast Extract Mannitol Agar) sown into lyophilization tubes containing skim milk, frozen at -85 °C and dehydrated at -55 °C with vacuum. According to the cultural counting results continued for 35 months after lyophilization, while the living cells in R11 strains and R12 strains found as 41x109 and 35.5x109 cell mL-1, respectively, the best result was determined in the R13 strain as 51.5x109 cell mL-1. In the greenhouse trials, the dry weight of plant`s green parts, the number of nodules and total nitrogen amounts were evaluated. The tested strains were inoculated with bean varieties peculiar to themselves and the lyophilizated cultures gave similar results with the main cultures kept in tubes with YMA.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2008.2032.2035
URL: https://scialert.net/abstract/?doi=pjbs.2008.2032.2035
INTRODUCTION
Rhizobium bacteria are preserved under various conditions in order not to lose their nitrogen fixation and genetic change (Antheuniss, 1973; Miyamato-Shinogara et al., 2006). Since, the cultures used continuously in the production are preserved best in the leaning tubes of agar, it facilitates its reproduction (Antheuniss et al., 1981). However, when preserving cultures in the agar medium, it takes a lot of efforts to transfer them repeatedly into a new medium every other month (Abadias et al., 2001; Miyamato-Shinora et al., 2006). In the recent studies, performing the control of cultures` activeness and effectively nodulation has been reported to be essential (Safronova and Novikona, 1996; Willems et al., 2001). When compared to the other preservation methods, the lyophilization method (frozen and dehydrated) has been determined to be used successfully with a widely range (Abadias et al., 2001; Miyamato-Shinogara et al., 2000).
Although, there are different views on the preservation temperatures of frozen and dehydrated cultures, preservation at low temperatures is recommended by Miyamato-Shinogara et al. (2006), Malik (1990) and Revellin et al. (2000). Willems et al. (2001) has suggested that Rhizobium cultures are preserved at either 15 °C in a dry place or at 4 °C in the fridge. The vigor tests carried out after lyophilization are usually done in the cultural counting technique and the results are presented as the number of living cells per milligram (Peter and Reichart, 2001; Ekdawi-Sever et al., 2003). In the preparation of culture collections of Rhizobium, lyophilized cultures are reported to be preferred in the greenhouse and field trials (Miyamato-Shinogara et al., 2006).
This study has been carried out to constitute the long time preservation of native Rhizobium strains, whose activeness was determined in the earlier study (Küçük, 2004), isolated from the root nodules of bean varieties that are cultivated widely in Turkey, providing the supply of the essential materials of Turkey`s developing microbial inoculant production and the formation of Rhizobium cultural collections.
MATERIALS AND METHODS
The strains: In this study, Rhizobium R11, R12 and R13 strains, whose activeness was determined from earlier study (Küçük, 2004), were used.
The preparation of lyophilization process: The cultures been purified in Petri dishes with Yeast Extract Mannitol Agar (YMA) then transferred into medium containing 20% skim milk in lyophilization tubes (2 mL) (Malik, 1990). After the tubes having been frozen at -85 °C, they were dehydrated at -55 °C under vacuum (Jenning, 1999). The samples were accumulated at +4 °C.
The vigor tests: During the preservation, a vigor test was performed for the cultures every other month. The cultures were dissolved in the yeast extract mannitol broth at the time of counting (Ekdawi-Sever et al., 2003). At the end of three days development, the number of living cells after lyophilization was obtained by cultural counting technique (Schoung et al., 2006).
Greenhouse tests: The test subjects include lyophilizated R11, R12, R13 strains, inoculated and noninoculated subjects. The Rhizobium strains that defined as Rhizobium bacteria in the earlier study (Küçük, 2004) were inoculated with bean varieties having high nodulation and nitrogen fixation peculiar to themselves. (R11, R12, R13 strains inoculated with Akman 98, Göynük 98 and Şehirali 90 bean varieties, respectively). The bean seeds (Phaseolus vulgaris L.) were provided from Anatolian Agricultural Research Institute in Eskişehir, Turkey.The seeds were soaked into 95% sodium hypochloride solution for 5 min and their surface sterilization were made by washing using sterile distilled water (Daba and Haile, 2002).
Afterwards, the seeds were sown into pots containing sterile peat (3 g). After the lyophilization strains having been developed in yeast extract mannitol broth 10 mL of culture (109 cell mL-1) was added to developing roots. At the end of 35 mount development period, the roots sections of plants were cut and the numbers of nodules were examined by Mostasso et al. (2002) and Daba and Haile (2002).
The green part sections of the plants were dehydrated at 65 °C in the oven until they were with stable weight, then weighted and their nitrogen amounts were determined with Kjeldahl method. All parameters were statistically analyzed using an analysis of variance (Yurtsever, 1984).
RESULTS AND DISCUSSION
In the recent study, while lyophilizated bacteria cultures were preserved at +4 °C, the vigor test was carried out every other month for 35 months. At the end of 35th month, nodulation and nitrogen fixation activity in the tested bean varieties were determined. The living Rhizobium numbers obtained from culture counting after the lyophilization process performed every other month are shown in Table 1. The starter values obtained after lyophilization varied between 263.5x109 and 35.5x109 cell number mL-1 (Table 1). The results in Table 1 show that, the existence of change on the living Rhizobium cell numbers performed every other month after the lyophilization process was determined. An analysis of variance of data indicated that strain, time, strainxtime (SxT) were significantly (0.01 level) (Table 1). Since the lyophilization is a method preventing the biological originated material from being spoiled, it is preferred in the Rhizobium microbiology to preserve the cultures for long time (Antheuniss, 1973; Miyamato-Shinogara et al., 2000, 2006; Safronova and Novikona, 1996; Willems et al., 2001; Abadias et al., 2001; Jenning, 1999).
| Table 1: | Colony forming units after freeze-drying (109 cell mL-1) |
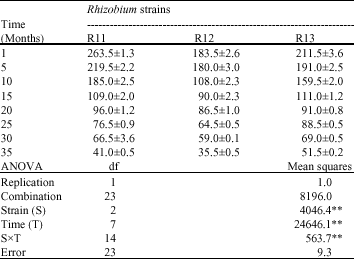 | |
| Significant at the **0.01 levels of probability, respectively | |
Miyamato-Shinogara et al. (2006), Willems et al. (2001) and Peter and Reichart (2001) showed that after lyophilization, the starter values being high like 109 cell mL-1 originated from their using intensive cell numbers at the start of lyophilization, taking possible bacteria loss number into account during the lyophilization process. In addition, in the performed culture counting after lyophilization every other month continued for 35 months, the decreasing vigor of Rhizobium strains when compared to their starter values, as Safronova and Novikona (1996) observed that the deaths occurred in time are thought to be originated from Rhizobium bacteria`s being quite sensitive to the dry environment.
In the studies carried by Safronova and Novikona (1996), in order to preserve Rhizobium bacteria, when they compared the methods of lyophilization with the method of keeping in the liquid nitrogen, they determined the lyophilization as the best preserving method for Rhizobium bacteria and also stated that the symbiotic characteristics of strains do not change at the end of the study.
The number of counted nodules of tested plants in their flowering period, the dry substance weight dehydrated at 65 °C and their total nitrogen extent are shown on Table 2. The highest nodule number which is R12 was obtained from inoculated Göynük 98 bean variety. Nodulation was not detected in the uninoculated beans. While the highest dry weight 2.90 g pot-1 was given by the inoculated with R12, the least dry weight 1.21 g pot-1 was given by the control (uninoculated) application. An analysis of variance of data indicated that, combination and treatment were significantly on nodule number (Table 2) and combination, strain, strainxtime were significantly on shoot dry weight (Table 2), combination, treatment and strain were significantly on N% (Table 2). The results in Table 2 show that, the total nitrogen varied between 83.43 and 130.5 mg plant-1. In the greenhouse trials carried out by Mostasso et al. (2002) and Daba and Haile (2002), the variation of the dry weights of bean plants being between 2.75 and 2.68 g pot-1 is harmonious with the our trial`s results.
| Table 2: | Effects of strains on bean varieties |
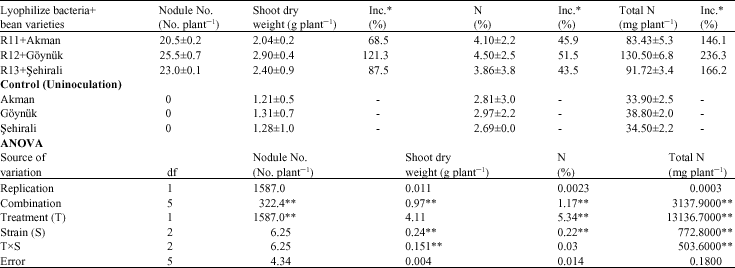 | |
| *Inc.: Increase, Significant at the **0.01 levels of probability, respectively | |
Consequently, according to the cultural counting performed after lyophilization, the starter values of lyophilizated cultures varied between 35.5x109 to 263.5x109 cell mL--1. Between three tested strains, the highest vigor value was obtained from R13. The values belong to lyophilizated cultures and the main cultures kept in tubes with YMA were found similar to each other. During the preservation of isolates, when taking some factors into consideration such as absence of contamination, having no mutations due to the continuous inoculated that happening continuously, having economically low cost, the ability of being preserved securely for a long time, using the lyophilization technique can be recommended for preservation of Turkish Rhizobium strains.
ACKNOWLEDGMENTS
This study supported by the Scientific and Technological Research Council of Turkey (TUBITAK-BAYG).
REFERENCES
- Abadias, I., A. Benabarre, N. Teixido, J. Usall and I. Vinas, 2001. Effect of freze drying and protectants on viability of the biocontrol yeast Candida sake. Int. J. Food Microbiol., 65: 173-182.
CrossRef - Antheuniss, J., 1973. Viability of lyophilized microorganisms after storage. Antonie van Leeuwenhoek, 39: 243-248.
CrossRef - Antheuniss, J., J.W. de Bruin-Tol and S.V. Pol-Van, 1981. Survival of microorganisms after drying and storage. Antonie van Leeuwenhoek, 47: 539-545.
CrossRef - Daba, S. and M. Haile, 2002. Effects of rhizobial inoculant and nitrogen fertilizer on yield and nodulation of common bean under intercropped conditions. J. Plant Nutr., 25: 1443-1445.
CrossRef - Ekdawi-Sever, N., L.A. Goentoro and J.J. de Pablo, 2003. Effects of annealing on freeze dried Lactobacillus acidophilus. J. Food Sci., 68: 2504-2511.
CrossRef - Küçük, Ç., 2004. Eskişehir bölgesinde yetiştirilen fasulyelerin (Phaseolus vulgaris L.) kök nodüllerinden izole edilen Rhizobium suşlarinin karakterizasyonu, simbiyotik özelliklerinin belirlenmesi ve bunlarin inokulant olarak kullanilabilirliğinin araştirilmasi OGÜ. Fen Bilimleri Enst. Eskişehir, Türkiye, Sayfa 189.
- Malik, A.K., 1990. A simplified liquid-drying method for the preservation of microorganisms sensitive to freezing and freze-drying. J. Microbiol. Methods, 12: 125-132.
CrossRef - Miyamato-Shinogara, Y., T. Imaizumi, J. Sukenobe, Y. Murakami, S. Kawamura and Y. Komatsu, 2000. Survival rate of microbes after freeze-drying and long term storage. Cryobiology, 41: 251-255.
CrossRef - Miyamato-Shinogara, Y., J. Sukenobe, T. Imaizumi and T. Nakahara, 2006. Survival curves for microbial species stored by freeze-drying. Cryobiology, 52: 27-32.
CrossRef - Mostasso, L., F.L. Mostasso, B.G. Dias, M.A.T. Vargas and M. Hungria, 2002. Selection of bean (Phaseolus vulgaris L.) rhizobial strains for Brazilian Cerrados. Field Crops Res., 73: 121-132.
CrossRef - Safronova, V.I. and I. Novikona, 1996. Comparison of two methods for root nodule bacteria preservation: Lyophilization and liquid nitrogen freezing. J. Microbiol. Methods, 24: 231-237.
CrossRef - Sakane, T. and K. Kuroshima, 1997. Viabilities old dried cultures of various bacteria after preservation for over 20 years and their prediction by accelerated storage test. Microbiol. Cult. Collect., 13: 1-7.
CrossRef - Schoung, A., J. Olsson, J. Carlfors, J. Schnurer and S. Hakanson, 2006. Freeze-drying of Lactobacillus coryniformis Si3-effects of sucrose concentration, cell density and freezing rate on cell survival and thermophysical properties. Cryobiology, 53: 119-127.
CrossRef - Revellin, C., G. Meunier, G. Sommer, P. Wadoux and G. Catroux, 2000. Changes in the physiological and agricultural characteristics of peat based Bradyrhizobium japonicum inoculants after long term storage. Applied Microbiol. Biotechnol., 54: 206-211.
Direct Link - Wendt, C., B. Dietze, E. Dietze and H. Ruden, 1997. Survival of Acinetobacter baumanni on dry surfaces. J. Clin. Microbiol., 35: 1394-1397.
Direct Link - Willems, A., B. Hoste, J. Tang, D. Jannssens and M. Gillis, 2001. Differences between subcultures of the Mesorhizobium loti type strain from different culture collections. Syst. Applied Microbiol., 24: 549-553.
CrossRef








