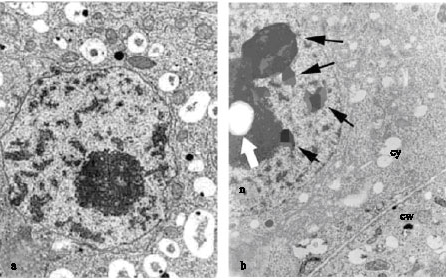
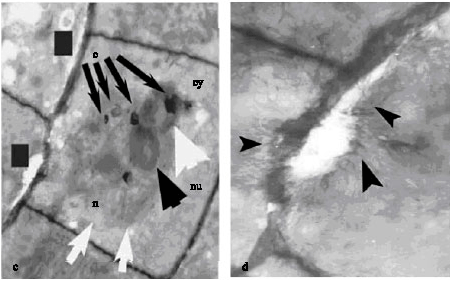
Research Article
Structural Changes in Root Tips of Wheat (Triticum aestivum L.) in Response to Olive Oil Mill Wastewater
Department of Biology, Faculty of Arts and Sciences, Trakya University, Edirne, Turkey
U. Sidal
Department of Biology, Faculty of Arts and Sciences, Muradiye Campus, Celal Bayar University, Manisa 45030, Turkey
G. Huseyin
Department of Pathology, Trakya University, Edirne, Turkey