Research Article
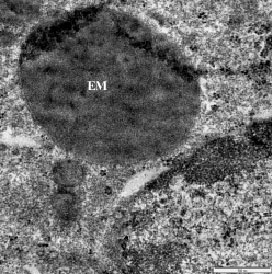
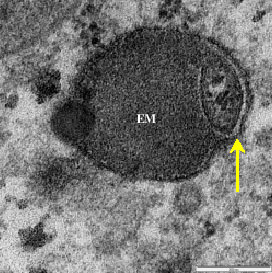
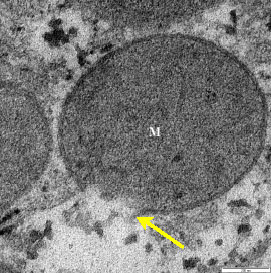
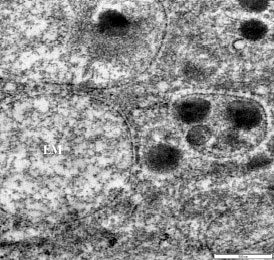
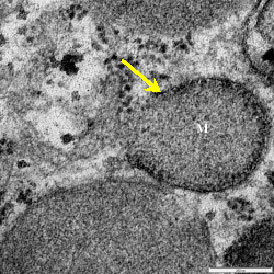
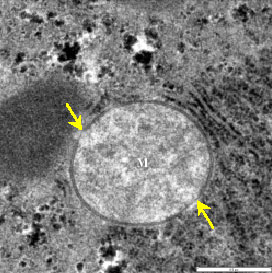
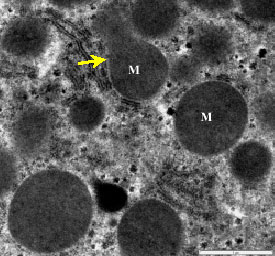
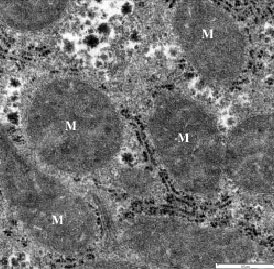
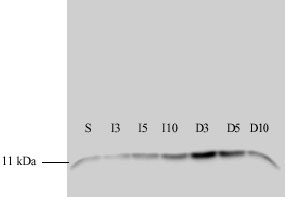
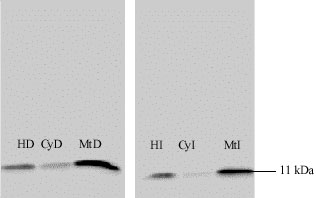
Morphological Alteration in Mitochondria Following Diclofenac and Ibuprofen Administration
Department of Human Anatomy,Faculty of Medicine and Health Sciences, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
S. Fakurazi
Department of Human Anatomy,Faculty of Medicine and Health Sciences, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
H. Ithnin
Department of Pathology, Faculty of Medicine and Health Sciences, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia