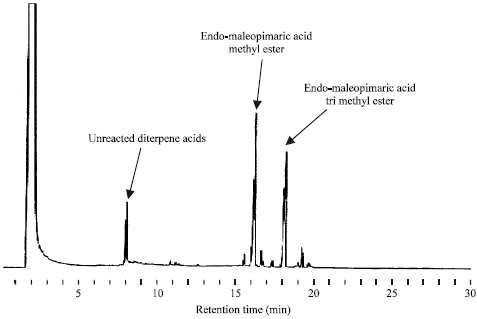
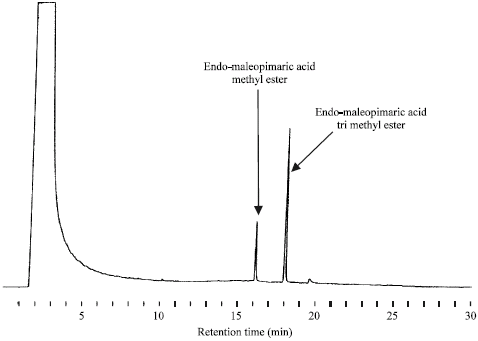
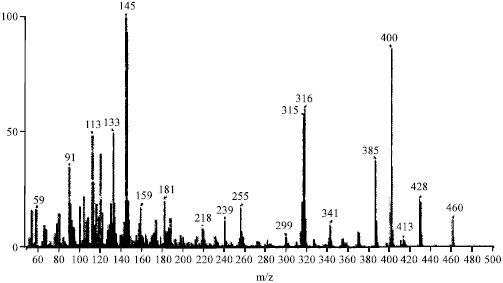
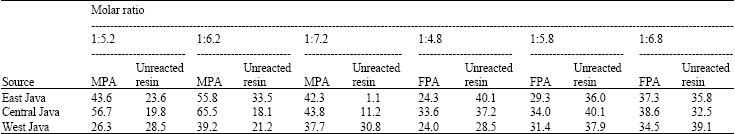
Research Article
Maleo- and Fumaro-Pimaric Acids Synthesized from Indonesian Pinus merkusii Rosin and Their Sizing Properties
Forest Products Research and Development Center, FORDA. Jl, Gunung Batu 5, Bogor, Indonesia
Sanro Tachibana
Faculty of Agriculture, Ehime University, 3-5-7 Tarumi, Matsuyama, Ehime, 790-8566, Japan