Research Article
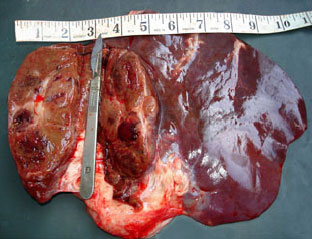
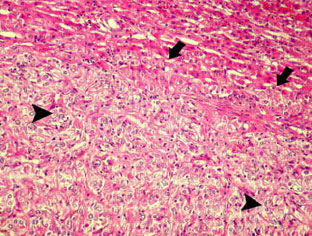
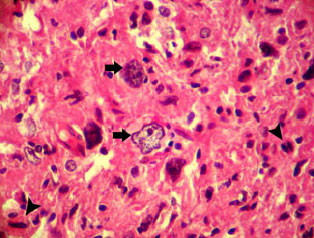
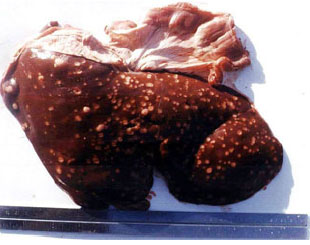
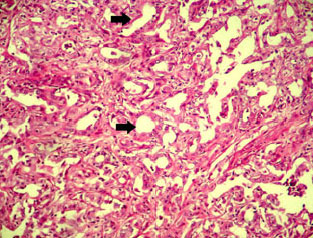
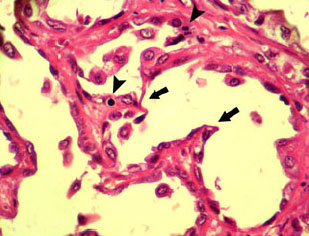
An Abattoir Study on Hepatic Tumors of Sheep
Department of Pathobiology, Faculty of Veterinary Medicine, Islamic Azad University-Tabriz Branch, Iran
A. Rezaie
Department of Clinical Sciences, Faculty of Veterinary Medicine, Islamic Azad University-Tabriz Branch, Iran
Gh. Mousavi
Department of Clinical Sciences, Faculty of Veterinary Medicine, Islamic Azad University-Tabriz Branch, Iran