ABSTRACT
This study is to inspect how the variation of molecular structures and functional groups present in our models, monoazo dye (Tartrazine) and diazo dye (Ponceau), affects decolorization capabilities of green algae, cyanobacteria and diatoms. The results revealed that the removal of azo dyes was rapid at the initial period of study (3 days) and became slowly with the time (6 days). The maximum decolorization was observed at 5 ppm Tartrazine with S. bijugatus (68%) and N. muscourm after 6 days incubation. The reduction of color removal appears to be related to the molecular structure of the dyes and species of algae used. The culture of the diatom Nitzschia perminuta was completely died after 2 days of incubation. Azo reductase of algae, which is responsible for degradation of azo dyes into aromatic amine by breaking the azo linkage, was estimated. IR spectra represented a new peak at 3300 cm-1 and a reduction in the azo band at 1642-1631 cm-1. In order to investigate the sorption behavior of algae, Langmuir equilibrium model was tested.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2008.1310.1316
URL: https://scialert.net/abstract/?doi=pjbs.2008.1310.1316
INTRODUCTION
Azo dyes are an important class of synthetic organic compounds used as coloring agents in the textile, paint, ink and plastic industries. Azo dyes are one of the oldest industrially synthesized organic compounds and represent the major group (60-70%) of the more than 10,000 dyes currently manufactured (Bras et al., 2005). However, large amounts of these dyes remain in the effect after the completion of the dyeing process. Consequently very small amount of dyes in waste water is highly visible (Marungruenga and Pavasant, 2006; Wang et al., 2005). A side from their negative aesthetic effects, certain azo dyes and their biotransformation products has been shown to be toxic to aquatic life and mutagenic to humans (Bafana et al., 2008; Brown and Hamburguer, 1987). Azo dyes are stable compounds, difficult to destroy or to be decomposed by common treatment in a biological treating station. Recently the application of algae has been receiving increasing attention in the field of waste water decolorization. Many researchers have studied the effect of algae and enzymes on decolorization characteristics since immobilization provide distinct stability over free cells. Algae have been studied in the field of decolorization of industrial effluents (Semple et al., 1999; Jianwei et al., 1997; Daneshvar et al., 2007a; Vijayaraghavan and Yun, 2008). This is due to the versatile ability of the algae to degrade, partially or completely various dyes. Azo dyes are characterized by the presence of one or more azo group (-N = N-) bonds to aromatic rings (Bafana et al., 2008) Azo dyes may be decolorized by cleavage of the azo bond, to which the color is associated (Wuhrmann et al., 1980). The reduction of azo dyes results in the formation of aromatic amines that mostly cannot be metabolized, with the exception of a few examples bearing hydroxyl and carboxyl groups, which can be fully degraded (Razo-Flores et al., 1996; Kudlich et al., 1999). Hence, the majority of azo dyes are mutagenic and carcinogenic to humans as well as other animals removal of hazardous industrial effluents is one of the growing needs of the present time. So, in this present work, experiments on the degradation of two azo dyes, a monoazo dye (Tartrazine) and a diazo dye (Ponceau) were carried out using green algae, diatom and cyanobacteria, to study the degradation of different azo dyes and hence to estimate the activity of azo reductase involved in the degradation of azo dyes. The azo reduction process was monitored by UV-visible spectrophotometry. In order to evaluate the removal capacity of algae, Langmuir isotherm studies were conducted.
MATERIALS AND METHODS
Azo dyes: Figure 1 showed the chemical structure of monoazo dye (Tartrazine) and diazo dye (Ponceau ss) used for decolorization study. They were obtained from Aldrich (Germany).
Algae and culture conditions: The algal strains were obtained from Algal Laboratory, Botany Department, Faculty of Science, Tanta University, Egypt. The green algal strains were Chlorella ellipsoidea, Chlorella kessleri, Chlorella vulgaris, Scenedesmus bijuga, Scenedesmus bijugatus and Scenedesmus obliquus; the cyanobacteria strains were Anabaena laxa, Anabaena subcylindrica, Nostoc muscourm and Oscillatoria angusta and the diatom was Nitzschia perminuta. The green algae were cultured in a sterile TAP (Tris-Acetate-Phosphate) medium (Harris, 1989). Allen medium (Allen, 1968) was used for cyanobacteria and diatom.
Decolorization study: The experiments were conducted in Erlenmeyer flasks containing algal cultures and azo dyes at different initial dye concentrations (5, 10 and 20 ppm). The cultures were incubated at 25°C for 6 days. The dye concentration was determined after 3 and 6 days of incubation by measuring the absorbance of the cell-free supernatent of the sample at the maximal absorption wave length (i.e., λmax). A sterile cell-free medium was chosen as the control. The changes in the dyes absorption spectra were recorded by using a UV-V is spectrophotometer (Shimazu UV-1601).
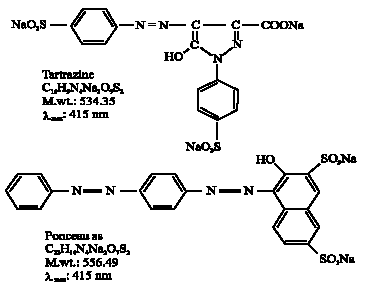 | |
| Fig. 1: | Chemical structures of the azo dyes used |
Azo reductase activity:The azo reductase preparation was made according to the producers described by Idaka et al. (1987 a,b ). The reductase activity of tested algae was studied after 3 and 6 days of incubation with 20 ppm azo dye concentration.
Infrared analysis (IR): The infrared spectra of the two azo dyes and the algae grown is dyes after the incubation period (6 days ) were analyzed using Perkin Elmer 1430 infrared spectrophotometer within the range 500-4000 cm-1.
RESULTS AND DISCUSSION
Azo dye decolorization: The degree of decolorization for monoazo dye (Tartrazine) and diazo dye (Ponceau) by some green algae, cyanobacteria and diatoms were studied at 5, 10 and 20 ppm dyes concentration for 3 and 6 days incubation. Figures 2 and 3 showed the decolorization (%) of different Tartrazine concentration by green algae. The results obtained that the amount of color removal varies with varying initial dye concentrations. The maximum decolorization was observed with 5 ppm azo dyes concentration and slightly decreased up to 20 ppm with both green algae and cyanobacteria. In this connection, Aydin and Baysal (2006) found that dye removal is highly concentration dependent and approximately attributed to bioconversion. Also, Bafana et al. (2008) found that the decolorization was found to be due to both biological azo dye reduction and adsorption. The increase of decolorization of azo dyes seems to be related to the molecular structures of the dyes (Jinqi and Houtian, 1992), adsorption to the algae (Chen et al., 2003) and rapid degradation of the dye (Daneshvar et al., 2007 a,b). The rapid decolorization of Tartrazine and Ponceau was observed after 3 incubation days and then become slow with the time. This was caused by strong attractive force between the azo dyes
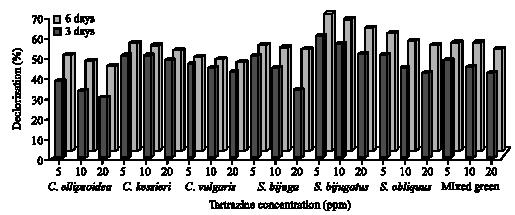 |
| Fig. 2: | Decolorization of different concentrations (5, 10 and 20 ppm) of Tartrazine by green algae after 3 and 6 days incubation |
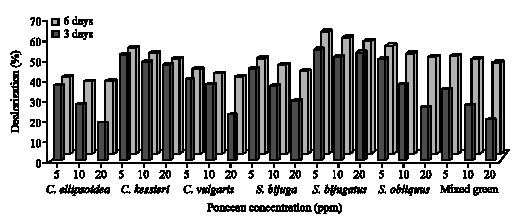 |
| Fig. 3: | Decolorization of different concentrations (5, 10 and 20 ppm) of Ponceau by green algae after 3 and 6 days incubation |
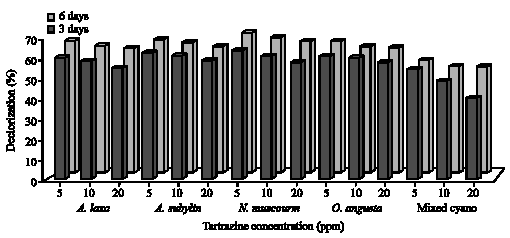 |
| Fig. 4: | Decolorization of different concentrations (5, 10 and 20 ppm) of Tartrazine by cyanobacteria after 3 and 6 days incubation |
molecules and the algae; fast diffusion onto the external surface was followed by fast diffusion into the algal cells to attain rapid equilibrium.
The extent of color removal of Tartrazine ranged from 29 to 68% in green algal cultures (Fig. 2), whereas in cyanobacteria ranged from 57 to 70% (Fig. 3) after 3 and 6 days of incubation. The algae S. bijugatus and N. muscourm were the highest removal for Tartrazine color. Several cyanobacterial strains posses to their outer membrane, additional surface substances mainly of polysaccharide nature (Bertocchi et al., 1990). De Philippis et al. (2001) surveyed the ability of cyanobacteria polysaccharide to remove pollutants from aqueous solutions and this may explain why the cyanobacteria strains were higher removal of color than the green algae. On the other hand, Ponceau decolorization by green algae ranged between 18 to 62% and between 23 to 59% with cyanobacteria after 6 incubation days (Fig. 4, 5). The maximal removal of Ponceau color was observed with S. bijugatus (55 and 62%) and N. muscourm (55 and 59%) at 5 ppm after 3 and 6 days incubation, respectively.
It was obious that diazo dye, Ponceau, was less decolorized in contrast to monoazo dye (Tartrazine). Chang et al. (2001) suggested that both partial reduction and complete cleavage of the azo bond could contribute to decolorization of azo dye. This result might be explained by Zimmermann et al. (1982) as follows the charged groups (sulfonic group) in the proximity to the azo group hinder the reaction (decolorization). This might clearly explain why Ponceau was not biodegradable compared to Tartrazine. Tartrazine might be easier to be transported (and/or) adsorbed into cellular compartment this because of the sulphonic groups to azo bond (-N = N-) in Tartrazine was a strong electron-with drawing groups through resonance to cause an enhanced of color removal to be easily decolorized (McMurry, 2004; Hsueh and Chen, 2007 ). Moreover, Chen (2002, 2006) and Sugiura et al. (1999) mentioned that diazo dye-Ponceau was not easily to be decolorized perhaps due to the persistent toxicity of azo bond(s) in Ponceau and or generated intermediates.
The results recorded a completely death for Navicula sp. at the different azo dyes concentrations after 2 days of
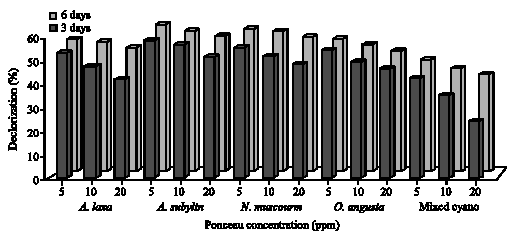 |
| Fig. 5: | Decolorization of different concentrations (5, 10 and 20 ppm) of Ponceau by cyanobacteria after 3 and 6 days incubation |
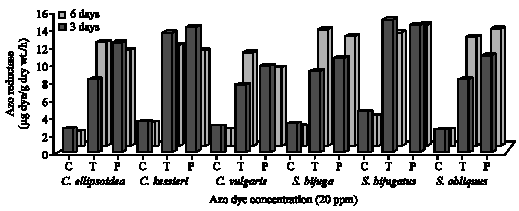 |
| Fig. 6: | Azo reductase activity (μg dye/g dry wt./h) of green algae under the effect of Tartrazine (T) and Ponceau (P) at the concentration 20 ppm after 3 and 6 days incubation. (C = Control) |
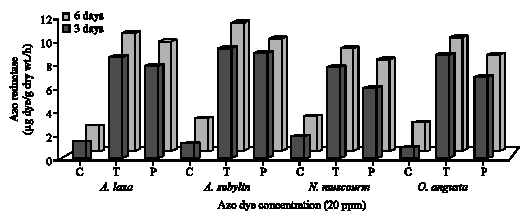 |
| Fig. 7: | Azo reductase activity (μg dye/g dry wt./h) of cyanobacteria under the effect of Tartrazine (T) and Ponceau (P) at the concentration 20 ppm after 3 and 6 days incubation (C = Control) |
incubation. Azo dyes may affect photosynthetic activity in aquatic life because of reduced light penetrate and may also be toxic to some aquatic life due to the presence of aromatics (Daneshvar et al., 2007 a).
Azo reductase activity: To study the azo reductase activity of tested algae, the concentration 20 ppm of Tartrazine and Ponceau was chosen to carry out their effect on the algal enzymatic activity after 3 and 6 days incubation. The activity of azo reductase for green algae and cyanobacteria was shown in Fig. 6 and 7. The results showed that the addition of azo dyes, Tartrazine and Ponceau, to the algal cultures induced azo dye reductase activity as compared with control. This may be due to the ability of algae to induce azo dye reductase activity under azo dye stress and these results are in agreement with that
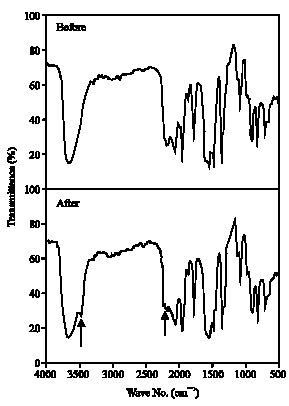 | |
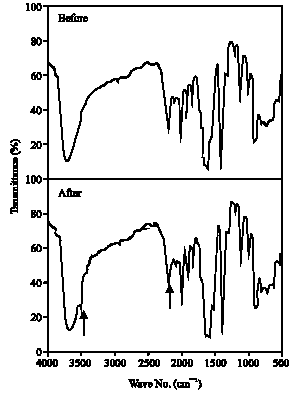
| Fig. 8: | Infrared spectrum of Tartrazine before and after algal treatment |
obtained by Urushigawa and Yonezawa (1977). In a general, the maximum azo reductase activity (μg dye g-1 dry wt./h) was recorded with green algae as compared with control and cyanobacteria after 6 days incubation. The maximum reductase activity was observed with the green algae S. bijugatus (15.2 and 14.2 after 3 days) and the cyanobacteria A. subcylindrica (10.6 and 9.2 after 6 days) during the treatment with Tartrazine and Ponceau, respectively. Jinqi and Houtian (1992) studied azo reductase activity of some algae and they found that the azo reductase of C. vulgaris is an induced enzyme and the substrate itself can act as a kind of inducer. Also, Pandy et al. (2007) reported that azo reductase can act in breaking clown of the azo bond as follows:
IR:Figures 8 and 9 showed infrared spectra of Tartrazine and Ponceau, respectively, to identify their structural variations, before and after algal treatment. The results showed a new absorption band at 3300 cm-1 due to stretching vibration of NH2 group which support the reduction of azo group (-N = N-) to amino group by azo reductase. At the same time, the absorption band of the azo bond within the range 1642-1631 cm-1 was decreased (as indicated by arrows in the figures). These results are consistent with the results obtained by Jinqi and Houtian (1992).
 | |
| Fig. 9: | Infrared spectrum of Ponceau before and after algal treatment |
Langmuir adsorption isotherm:Adsorption isotherm describes how dyes interact with algae and so are critical in optimizing the use of algae. In order to optimize the design of a sorption system to remove dyes from effluents, it is important to establish the most appropriate correlation for the equilibrium curves. In the present study, the experimental data of two azo dyes-algae equilibrium isotherms were compared using Langmuir isotherm. Langmuir sorption model served to estimate the maximum uptake values, where they could not be reached in the experiments. Langmuir isotherm is represented by the following equation (Chiou et al., 2004).
Ce/qe = (1/Q0 b) + (Ce/Q0) |
where, Ce is the equilibrium concentration of dye solution (residual) and qe is the adsorbed dye on the algal cells. Q0 and b are constants related to adsorption capacity and rate of adsorption, respectively. Values of Q0 and b for the adsorption of dyes onto algal cells were calculated from the slope and intercept plot of Co/qe vs Ce. The essential features of the Langmuir isotherm can be expressed in terms of the dimensionless constant separation factor R which is defined by the following relationship given by Magdy and Daifullah (1998):
R = 1/(1+bC0) |
| Table 1: | Langmuir isotherm constants for the decolorization of Tartrazine and Ponceau by green algae and cyanobacteria |
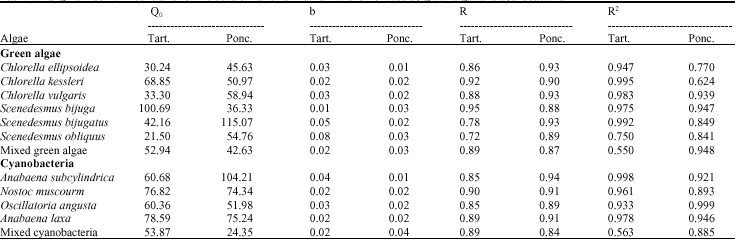 | |
The isotherm of Tartrazine and Ponceau were found to be linear over the whole concentration range studies and the correlation coefficients (R2) were extremely high as shown in Table 1. The R2 was less than 1 and greater than zero indicating that tested algae are favorable for adsorption of azo dyes.
CONCLUSIONS
The presence of dyes imparts an intense color to effluents, which lead to environmental problems. It may be concluded that algae undoubtedly have the potential to rapidly, efficiently and effectively remove dyes to low concentrations. Moreover, this biosorption process could be adopted as a cost effective and efficient approach for decolorization of effluents ant it may be an alternative to more costly materials.
REFERENCES
- Allen, M.M., 1968. Simple conditions for growth of unicellular blue-green algae on plates. J. Phycol., 4: 1-4.
CrossRefDirect Link - Aydin, H. and G. Baysal, 2006. Adsorption of acid dyes in aqueous solutions by shells of bittim (Pistacia khinjuk stocks). Desalin, 196: 248-259.
CrossRefDirect Link - Bafana, A., K. Krishnamurthi, S. Devi and T. Chakrabarti, 2008. Biological decolourization of C.I. direct black 38 by E. gallinarum. J. Hazard. Mater., 157: 183-193.
CrossRefPubMedDirect Link - Bertocchi, C., L. Navarini, A. Cesaro and M. Anastasio, 1990. Polysaccharides from cyanobacteria. Carbohyd. Polym., 12: 127-153.
CrossRef - Brown, D. and B. Hamburguer, 1987. The degradation of dyestuffs. Part 3. Investigation of their ultimate degradability. Chemosphere, 16: 1539-1553.
CrossRef - Chang, J.S., C. Chou, Y.C. Lin, P.J. Lin, J.Y. Ho and T.L. Hu, 2001. Kinetic characteristics of bacterial azo-dye decolorization by Pseudomonas luteola. Water Res., 35: 2841-2850.
CrossRefDirect Link - Chen, B.Y., 2002. Understanding decolorization characteristics of reactive azo dyes by Pseudomonas luteola: Toxicity and kinetics. Process Biochem., 38: 437-446.
CrossRefDirect Link - Chen, K.C., J.Y. Wu, D.J. Liou and S.C.J. Hwang, 2003. Decolorization of the textile dyes by newly isolated bacterial strains. J. Biotechnol., 101: 57-68.
CrossRefDirect Link - Chiou, M., P. Ho and H. Li, 2004. Adsorption of anionic dyes in acid solutions using chemically cross-linked chitosan beads. Dyes Pigment, 60: 69-84.
CrossRef - Daneshvar, N., M. Ayazloo, A.R. Khataee and M. Pourhassan, 2007. Biological decolorization of dye solution containing Malachite Green by microalgae Cosmarium sp. Bioresour. Technol., 98: 1176-1182.
CrossRefDirect Link - Daneshvar, A., A.R. Khataee, M.H. Rasoulifard and M. Pourhassan, 2007. Biodegradation of dye solution containing Malachite Green: Optimization of effective parameters using Taguchi method. J. Hazar. Mater., 143: 214-219.
CrossRef - De Philippis, R., C. Sili, R. Paperi and M. Vincenzini, 2001. Exopolysaccharide-producing cyanobacteria and their possible exploitation: A review. J. Applied Phycol., 13: 293-299.
CrossRef - Hsueh, C. and B. Chen, 2007. Comparative study on reaction selectivity of azo dye decolorization by Pseudomonas luteola. J. Hazar. Mater., 141: 842-849.
CrossRef - Idaka, E., T. Ogawa and H. Horitsu, 1987. Reductive metabolism of aminoazobezene by Psudomonas cepacia. Bull. Environ. Contam. Toxicol., 39: 100-107.
CrossRef - Idaka, E., T. Ogawa and H. Horitsu, 1987. Oxidative pathway after reduction of p-aminoazobenzen by Pseudomonas cepacia. Bull. Environ. Contam. Toxicol., 39: 108-113.
CrossRef - Jinqi, L. and L. Houtian, 1992. Degradation of azo dyes by algae. Environ. Pollut., 75: 273-278.
CrossRef - Magdy, Y.H. and A.A. Daifullah, 1998. Adsorption of a basic dye from aqueous solutions onto sugar-industry-mud in two modes of operations. Wast. Manage., 18: 219-226.
CrossRef - Marungruenga, K. and P. Pavasant, 2006. Removal of basic dye (Astrazon Blue FGRL) using macroalga Caulerpa lentillifera. J. Environ. Manage., 78: 268-274.
CrossRef - Pandey, A., P. Singh and L. Iyengar, 2007. Bacterial decolorization and degradation of azo dyes. Int. Biodeterior. Biodegrad., 59: 73-84.
CrossRefDirect Link - Razo-Flores, E., B. Donlon, J. Field and G. Letting, 1996. Biodegradability of N-substituted aromatics and alkylphenols under methanogenic conditions using granular sludge. Water Sci. Technol., 33: 47-57.
CrossRef - Semple, K.T., R.B. Cain and S. Schmidt, 1999. Biodegradation of aromatic compounds by microalgae. FEMS. Microbiol. Lett., 170: 291-300.
CrossRef - Sugiura, W., T. Miyashita, T. Yokoyama and M. Arai, 1999. Isolation of azo dye-degrading microorganisms and their application to white discharge painting of fabric. J. Biosci. Bioeng., 88: 577-581.
PubMedDirect Link - Vijayaraghavan, K. and Y. Yun, 2008. Biosorption of C.I. Reactive Black 5 from aqueous solution using acid-treated biomass of brown seaweed Laminaria sp. Dyes Pig., 76: 726-732.
CrossRef - Wang, S., Y. Boyjoo, A. Choueib and Z.H. Zhu, 2005. Removal of dyes from aqueous solution using fly ash and red mud. Water Res., 39: 129-138.
CrossRefDirect Link - Wuhrmann, K., K. Mechsner and T. Kappeler, 1980. Investigation on rate-determining factors in the microbial reduction of azo dyes. Eur. J. Applied Microbiol. Biotechnol., 9: 325-338.
CrossRefDirect Link - Zimmermann, T., H.G. Kulla and T. Leisinger, 1982. Properties of purified orange II azoreductase, the enzyme initiating azo dye degradation by Pseudomonas KF46. Eur. J. Biochem., 129: 197-203.
CrossRefDirect Link








