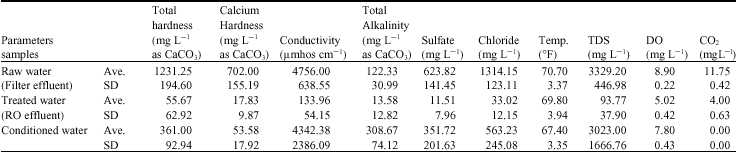
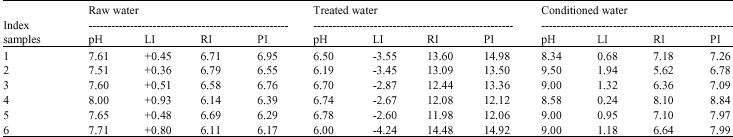
Research Article
Study for Determination of Industrial Water Corrosivity in Kashan Fajre Sepahan Galvanizing Mills During 2005-2006 Iran
Scientific Member of Kashan Medical Sciences University, Iran
M.B. Miranzadeh
Scientific Member of Kashan Medical Sciences University, Iran
A. Ahmadi Motlagh
Environmental Health Expert, Iran