Research Article
Production and Characterization of a Mesophilic Lipase Isolated from Bacillus stearothermophilus AB-1
Department of Botany and Microbiology, Faculty of Science, Helwan University, Ain Helwan, Cairo, Egypt
INTRODUCTION
Lipase (triacylglycerol acylhydrolase, E.C.3.1.1.3) is one of the well-known industrial useful enzymes, because it can catalyze different reactions including hydrolysis of triglycerides, transesterification and chiral synthesis of esters under natural conditions. Based on the versatile reaction properties of lipases, they have been widely used in industrial applications, such as food, chemical, pharmaceutical, detergent industries and organic synthesis (Schmidt-Dannert, 1999; Park et al., 2005; Gupta et al., 2006). Although lipases are widely found in animals, plants and microbes; microbial lipases have gained special industrial attention because of their versatility and availability. Many microorganisms are known as good producers of extracellular lipases (Ratledge and Tan, 1990; Eom et al., 2006). It was reported that strains of bacteria and yeast screened, the genera Pseudomonas, Bacillus, Candida, Rhodococcus and Staphylococcus grew on the waste oils and produced lipolytic activity. The highest producers were Pseudomonas sp. and Pseudomonas aeruginosa (Haba et al., 2000).
Also, screening for the presence of lipase activity in lactobacilli isolated from chouriço, a traditional Portuguese dry fermented sausage, a strain of Lactobacillus plantarum was chosen for extracellular lipase characterization and purification (Lopes et al.,2002). Moreover, a high-yield purification of an organic solvent-tolerant lipase from Pseudomonas sp. strain was obtained. The lipase exhibited the highest activity in the presence of palm oil as a natural oil and triolein as a synthetic triglyceride (Rahman et al., 2005). The ability of Thermomyces lanuginosus wild strains isolated from biohumus, mushroom and garden composts, decayed leaves, hazelnuts and raw coffee beans to hydrolyze synthetic (tributyrin, Tween 20, Tween 40, Tween 60 and Tween 80) and natural fatty substrates (sunflower, soybean, rapeseed and corn oil) was evaluated and whether the lipolytic activity depended on the isolation source was determined. The highest mean activity index was noted after growth on sunflower oil, followed by soybean oil and tributyrin; indices on other fatty substrates were low. Strains isolated from raw coffee beans showed the highest mean index, followed by those from biohumus and garden compost; the lowest index being for strains isolated from hazelnuts. Thus, the lipolytic activity index depended on the specific fatty substrate and the source of the isolates (Janda, 2005). Interestingly, strains of Halobacteria from an Algerian culture collection were screened for their lipolytic activity against p-nitrophenyl butyrate (PNPB) and p-nitrophenyl palmitate (PNPP). The results showed a clear correlation between salt and temperature requirement (Bhatnagar et al., 2005).
However, the enormous interests in thermophilic lipase were belonged to those generated from thermophlic organisms. Consequently, there is a steadily increasing demand to identify and characterize new lipases from different habitat (air, soil, water). The knowledge of mesophilic lipolytic enzymes in industrial applications is increasing at a rapid and exciting rate. Unfortunately, the studies on mesophilic lipases are incomplete and meager. Thus far, no attempt has been undertaken to organize this information.
In this study, a Bacillus AB-1 was isolated from air area around the laboratory. We expanded our researcher more specifically with respect to high production and novel properties of lipase from this strain, which was selected as the only lipase producer among bacterial strain tested. This study describes the production, purification and preliminary characterization of an extracelluar mesophilic lipase from the Bacillus stearothermophilus AB-1 for potential industrial application.
MATERIALS AND METHODS
Chemicals and reagents: Tributyrine stock emulsion was prepared in a ratio of tributyrine oil to gum Arabic (V/W) was (1/1). The emulsion was prepared by grinding thoroughly in a mortar 2 g of gum Arabic with 2 mL of fresh tributyrine oil. The mixture was then dissolved in 250 mL warm distilled water, small aliquot of the stock emulsion was done and stored in -20°C (Maia et al., 1999).
Organism and culture condition: Basal medium (BM), pH 7.0 was used to optimize the growth of the isolated strain AB-1 according to the method previously described by Maia et al. (1999) with slight modification as follow:
BM1 (g L-1 of deionized water): Tributyrine emulsion 2.0 mL; KHPO4, 1; NaNO3, 3; MgSO4, 0.5; peptone, 30; FeSO4.5H2O, 0.004 and Agar, 20.
BM2 (g L-1 of deionized water): Tributyrine emulsion 2.0 mL; KHPO4, 1; MgSO4, 0.5; ZnSO4, 4; FeSO4,1; MnSC4, 2; Yeast extract; 5 and Agar, 20.
BM3 (g L-1 of deionized water): Tributyrine emulsion 2.0 mL; Nutrient broth supplemented with CaCl2, 1; Agar, 20.
Strains show clear zone on plates were selected as candidate for further study.
| Table 1: | Identification of the strain AB-1 |
 | |
| +: Positive, -: Negative, NI: Not Identified | |
Identification of bacterial isolate: The Bacillus stearothermophilus AB-1 used in this study was isolated from the air by enrichment culture using Tributyrine as a substrate and identified according to the procedures described in Bergey`s Manual of Systematic Bacteriology (Claus and Berkeley, 1986) (Table 1).
Fermentative production of lipase by Bacillus stearothermophilus: The fermentative production of lipase was carried out on BM1 as best medium for production of lipase. In order to determine the optimal medium composition, various physical and chemical conditions were used, including different incubation periods, shaking, carbon and nitrogen sources, temperature and pH. Briefly, aliquots of 10 mL of this culture grown for 2 days withdrawn, centrifuged at 8000 rpm for 10 min. The supernatants were used to measure lipase activity, protein content and pH. The results were calculated from three independent cultures. Each measurement was carried out in triplicates.
Lipase production: The bacterial culture was grown in 250 mL Erlenmeyer flasks containing 50 mL of fermentation media inoculated with 1% (V/V) (0.5 OD600) of cell suspension and incubated at 35°C on a rotary shaker (120 rpm) for 48 h.
Determination of enzyme activity: Extracellular lipase activity was measured in culture supernatants after centrifugation (8000 rpm for 20 min) as described previously (Grbavcic et al., 2007) using Tributyrine emulsion as substrate. For each test run, a blank control was separately performed in each case. The reaction was stopped with methanol immediately after the addition of supernatant to the assay mixture. One unit was defined as the enzyme required for releasing one μmol of fatty acid per min under the assay conditions.
Determination of protein concentration: The protein concentration was determined according to Lowry method using bovine serum albumin as standard (Lowry et al., 1951).
Purification and characterization of the lipase Ammonium sulphate fractionation: The chart as mentioned by Dixon and Webb (1964) was applied to calculate the solid ammonium sulphate to be added to achieve any given concentration of the cell free filtrate under investigation. In order to partially purify the lipase enzyme, the filtrate was treated with 20, 40, 60, 80 and 100% saturation. For each Ammonium sulphate concentration, both the enzyme activity and protein content were measured and then the specific activity for each fraction can be calculated from the following equation.
Dialysis:Dialysis was carried out to remove the traces of (NH4)2SO4. The procedure was performed by introducing the previous precipitate in solution after dissolving in phosphate-buffer (pH 7.0), into a special plastic bag (10.000 Da) cut off. For dialysis against distilled water for 12 h, the obtained enzyme preparation was concentrated against sucrose to be available for the next application, column chromatographic technique.
Column chromatography Applying on sephadex G-200: The concentrated solution was applied to a column packed with sephadex G-200 equilibrated with (PPS, pH 7.0) and eluted with the same buffer. Preparation of the gel column and the fractionation procedure was carried out as mentioned by Soliman (2003). Fractions of 3.0 mL were harvested every 15 min and assayed for protein and lipase activity. Fractions with high lipase activity were pooled and concentrated. The samples were then stored at 4°C pending uses.
Thermostability: For the thermostability determination, the purified lipase (300 U mL-1) was incubated at different temperatures ranging from 25 to 55°C for 1 h in 20 mM phosphate buffer (pH 7.5) and the residual activity was measured as aforementioned above. Relative activity is expressed as a percentage of the maximum activity left under the experimental conditions.
pH stability:To measure the pH stability, the enzyme (300 U mL-1) was incubated at 35°C for 1 h in different buffers (with various pH). The residual activity was then measured.
RESULTS AND DISCUSSION
Preliminary study: The samples were collected from air using BM medium with different compositions named; BM1, BM2 and BM3 by gravity collection method. The isolate showing clear zone (lipolytic activity) around the bacterial growth in the plates is an indication for the production of extracellular lipase. An isolate showing the highest hydrolysis of Tributyrine (as substrate) in BM1 agar plates was chosen as a candidate for further characterization. Based on the cell shape, cell arrangement, relation to oxygen, physiological and biochemical tests; the isolate was identified as Bacillus stearothermophilus (Table 1).
Optimization of the composition of the culture medium: The lipase activity of Bacillus stearothermophilus was 300 U mL-1 under standard conditions. Study of the time-course lipase production showed that the highest lipase production with the 316 U mL-1 was observed 48 h after incubation with shacking (Fig. 1). The optimal incubation temperature for lipase production was determined to be 35°C when tested under the range of 25 to 55°C. Lipase production at 35°C was almost twice higher than that at 25 and 30°C (Fig. 2).
The pH of the culture is one of the most important environmental parameters affecting microbial cell growth and biochemical metabolism. Most natural environments have pH values between 5 and 9 and organisms with optimal pH in this range are most common. The lipase production increased dramatically up to 8 and dropped significantly at pH 9.
The optimal pH for lipase activity was pH 7.5 with a 3.9-fold higher than the pH 6 (Fig. 3), indicating that the extracellular lipase of Bacillus stearothermophilus was alkaline in nature. Since the lipase from Bacillus stearothermophilus AB-1 was found to be an alkaline lipase, the effect of important nutritional factors on the
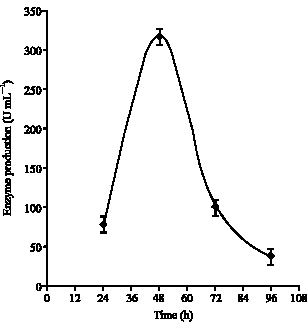 | |
| Fig. 1: | Effect of different incubation periods on the production of lipase by strain AB-1 |
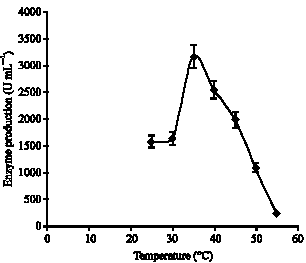 | |
| Fig. 2: | Effect of various temperatures on the production of lipase by strain AB-1 |
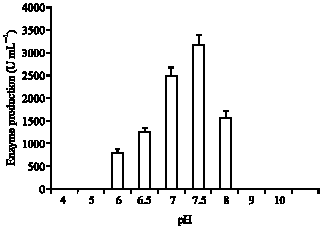 | |
| Fig. 3: | Effect of various pH on the production of lipase by strain AB-1 |
| Table 2: | Effect of various nitrogen sources on the production of lipase by strain AB-1 |
 | |
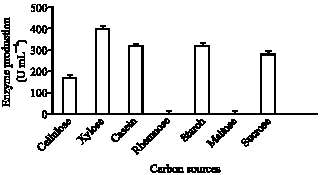 | |
| Fig. 4: | Effect of various carbon sources on the production of lipase by strain AB-1 |
production of the lipase was determined. Thus, 1% (w/v) of several different types of carbon sources was supplied (Fig. 4), in all cases other previously mentioned optimal conditions were taken into consideration. The strain grew on the carbon sources tested, yet monosaccharide, including Xylose was better than disaccharides, or polysaccharides for enzyme production. Although higher enzyme production of some lipid metabolizing enzymes on glycerol and glucose medium has been previously reported (Hass and Bailey, 1993; Shon et al., 1994), glycerol did not enhance the production of lipase in the current strain. The explained reason for the inhibitory effect of glycerol on lipase production is that glycerol was a product of the hydrolysis of triacylglycerols and it might decrease lipase production.
In addition, the effects of 0.1% (w/v) of various nitrogen sources on the enzyme production were also evaluated. As shown in Table 2, a higher enzyme production was observed in nitrogen sources; including alanine, phenylalanine, tryptophan and potassium nitrate.
Purification of lipase: In order to partially purify the lipase enzyme, the filtrate was treated with 20, 40, 60, 80 and 100% saturation. Present results showed that 60% ammonium sulphate was the best proportion, providing a higher enzyme activity, the finding which is consistent with that reported by Kanwar et al. (2002).
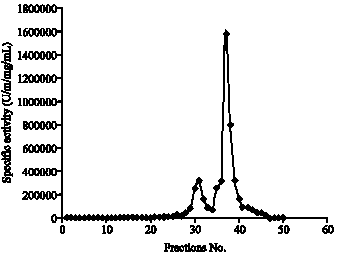 | |
| Fig. 5: | Purification of lipase by column chromatography technique |
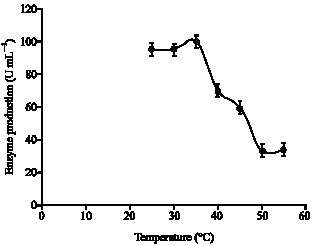 | |
| Fig. 6: | Effect of various temperatures on the stability of the purified lipase produced by strain AB-1 |
The precipitated lipase protein was separated and dialyzed against distilled water. The concentrated solution was applied to Sephadex G-200 gel filtration column equilibrated with phosphate buffer pH 7.5 and eluted with the same buffer. Fractions with high lipase activity were pooled and concentrated. The samples were stored at 4°C for further characterization (Fig. 5).
Effect of temperature on stability: The sephadex G-200 samples were held at a required temperature at pH 7.5 (Fig. 5). After incubation, the temperature was brought to 35°C and the residual activity of the enzyme was determined. The purified enzyme was stable at 35 and 40°C, 70% of its activity was retained (Fig. 6).
Effect of pH on stability: Stability of the crude and purified enzyme was investigated in buffer solutions of various pHs. The enzyme solution was incubated at 35°C for 1 h
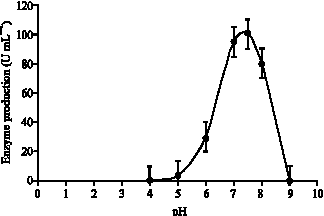 | |
| Fig. 7: | Effect of various pH on the stability of the purified lipase produced by strain AB-1 |
and the residual activity was determined (Fig. 7). Results showed that the enzyme is most stable at pH 7-8, retaining 80% of its activity at pH 8.
It has been reported that, Bacillus stearothermophilus secreted a thermostable lipase into the extracelluar medium, which was active at a temperature ranging from 60 to 65°C and a pH from 9 to 10 (Kim et al., 1997, 1998). To date a large number of Bacillus strains have been isolated for industrial applications (Horikoshi, 1971; Fritze et al., 1990; Khyami-Horani, 1996). Based on the above mentioned results we may suggest that isolate AB-1 could be identified as a new strain of Bacillus stearothermophilus, which differs from the strain of Nazina et al. (2001) in being mesophilic and slightly alkaline. The characteristic point of strain AB-1 is the production of alkaline mesostable lipase (300 U mL-1), which is optimally active at pH 7.5-8 and thermostable at 35-40°C. These characteristics distinguish the lipase produced by strain AB-1 from the other alkaline lipases produced by other bacilli previously reported by Moller et al. (1991), Lesuisse et al. (1993), Khyami-Horani (1996) and Kim et al. (1997, 1998). Addition of Xylose or starch to the growth medium, induce lipase production significantly, whereas, glucose and glycerol inhibited the formation. The inhibition of lipase formation by glucose was reported in earlier studies in microbial lipase (Satomura et al., 1958; Yamada et al., 1963).
The mesophilic lipase enzymes along with the producing microorganisms cover a broad spectrum of biotechnological applications. They include additives in detergents (cold washing), additives in food industries (fermentation, cheese manufacture, bakery, meat tenderizing), environmental bioremediations (digesters, composting, oil degradation or xenobiotic and/or molecular biology application).
Best to our knowledge, this the first report isolating Bacillus stearothermophilus AB-1 from the air. Moreover, the lipase enzyme produced by this strain is different from those previously described. Future plan, shall include gene cloning, sequencing and expression for industrial applications.
ACKNOWLEDGMENT
The author would like to thank Prof. Dr. A.M. Abd El-Aty for comments on the manuscript.