ABSTRACT
This research was carried out to determine if dominant morphological traits can be use instead of the rust resistance tests as marker characters for some agro-morphologic traits both leaf (Puccinia recondita Roberge ex Desmaz. f. sp. tritici.) and yellow rust (Puccinia striiformis Westendorp f. sp. tritici) disease. For this, 13 wheat parent (4 are T. durum Desf. and 9 are T. aestivum L.) were used and three main agro-morphologic traits (Namely, Leaf Colour (LC) (Dark and light green, Dg and Lg, relatively); Auricle Colour (AC) (White and Red, Wh and Re, relatively) and Leaf Waxiness (LW) (Waxy and Non waxy, Wx and Nw, relatively) were investigated at the seedling stage. They were found as dominant and concluded that investigated traits could be used as a morphological marker character for the leaf and yellow rust disease resistance tests under greenhouse conditions without doing complex and risky rust tests. And determined that, these traits may be use effectively without doing the complex rust tests in the limited populations or for the pre-selections. Doing with this, it will be bring a great accuracy, successful, correction and speed to the rust disease studies and provides important and valuable information for the plant breeders and pathologists. In addition, knowing of the resistance type (genetic mechanism etc.) in wheat facilitates it will be make to find out the suit and most appropriate breeding and selection methodology/methodologies.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2008.1.16
URL: https://scialert.net/abstract/?doi=pjbs.2008.1.16
INTRODUCTION
Wheat (Triticum sp.) plant is one of the second domesticated food crops after the barley and 8000 years it has been the basic staple food of the major civilizations of Europe, West Asia and North Africa (Vavilov, 1951). Today, wheat cultivation is being done on 614 mt ha-1, 149 mt production and 633 kg ha-1, yield level in the world (Anonymous, 2004). According to statistical data, world population was projected to be 7,9 billion people at the end of 2025 (Anonymous, 2005) and now, it is 6,606,008,519 billion (Anonymous, 2006). In simplistic terms and assuming little or no change in world per person consumption of wheat, a projection of 786 mt of wheat will be required annually for human use in the year 2025. If population growth continues to double the growth of wheat production, there will likely be serious difficulties in maintaining a wheat food supply for future generations. It is estimated that more than $5 billion are lost to cereal rusts each year and they have probably been a serious problem since the first cereal crops were grown in the Fertile Crescent (Hogenboom, 1993). On the other hand, the narrow genetic basis of durum and common wheat is a major obstacle for the improvement of these crops (Feldman and Sears, 1981) but in any case, it has a great importance to widen the genetic variation of desirable traits, particularly those affecting yield or quality. Resistance genes transferred from diploid to tetraploid and hexaploid levels generally confer decreasing levels of resistance (Roelfs et al., 1992). With the discovery of the genetic basis of resistance by Biffen (1905), physiological specialization in rust pathogens by Stakman and Levine (1962) and gene-for-gene interaction(s) by Flor (1956), the utilization of the hypersensitive (race-specific) resistance has dominated in wheat improvement (Curtis et al., 2002). The most economical and environmentally safe method to reduce the losses is the cultivation of resistant wheats, rust diseases can severely reduce wheat yields when susceptible cultivars are planted and conditions suit for a epidemy. Thus, resistance genes from the relatives of wheat genetic reservoir and offer new opportunities for maintaining diversification of the resistance gene combinations in new wheat cultivars to meet changes in the genes for virulence in rust populations (Knott and Bai, 1994). The aim of this study was to obtain more detailed information (especially in limited populations) based on visible and effective the selection for the leaf and yellow wheat rust resistances before doing the complex disease tests which consume more time and labour.
MATERIALS AND METHODS
Nine susceptible (AkOz 867, KOse 220/39, Kunduru 1149, Kunduru 414/44, Penjamo 62, Sertak 52, Sivas 111/33, Surak 1593/51 and Yektay 406) and 4 resistant wild genotypes (T. carthlicum, T. dicoccum, T. spelta and T. vavilovii) were used and 18 crosses (hybrids) were derived from them (Table 1). These cultivars and genotypes were sown and growth at the experimental fields of University of Ankara, Faculty of Agriculture, Department of Field Crops, (39°-36° N, 32°-40° E, asl 860 m), Ankara, Turkey in 1997-1998 and 1998-1999. Each plot consist of 5 rows, 1.0 m long and 0.20 m spaced, with the depth of 5.0-6.0 cm in a Randomized Block Design (RBD) with four replications. All plots were surrounded with a durum wheat cultivar çakmak 79. Recommended fertilizer, seeding rates and other agronomical practices were followed and no fungicide or growth regulator were used. Plots were irrigated with overhead sprinklers at four times during the boot stage (Zadoks 4.3-5.5) (Zadoks et al., 1974). Next year, plants were hybridized in the same way under Central Anatolian field conditions.
Rust tests: The seedlings were tested in a rust-free growth chambers under the greenhouse conditions, separately from each other under the controlled light and temperature conditions at a diurnal temperature cycle of 10-25°C and they were grown at a daily photo-period of 18 h, including 12 h at a maximum light intensity (24000 lux combined fluorescent and incandescent light, 20/15°C day/night temperature) and stepwise light increase and decrease, respectively, for one each (Roelfs et al., 1992). All seedlings were uniformly inoculated at the two-leaf stage with the population of urediospores of a leaf and stem rust races, suspended in a light mineral oil, Soltrol 170 (Phillips, Petroleum Co., Bartlesville, Oklahama), collected (Ozgen et al., 2004) from the Central Anatolian in Turkey. Then, plantlets were placed in a dew chamber with a humidifier for about at 9°C for 18 h dark period, followed by a stepwise increase in light and temperature. Afterwards, plants were then maintain at a low temperature profile of 15°C day and night as possible as
| Table 1: | Genotypes, scientific names, genome, chromosome numbers and responses to the leaf and yellow rusts |
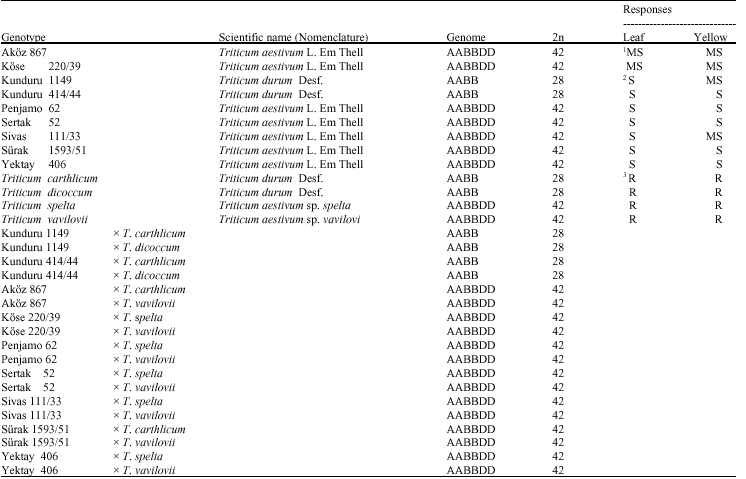 | |
| 1:Moderately Susceptible, 2: Susceptible, 3: Resistant | |
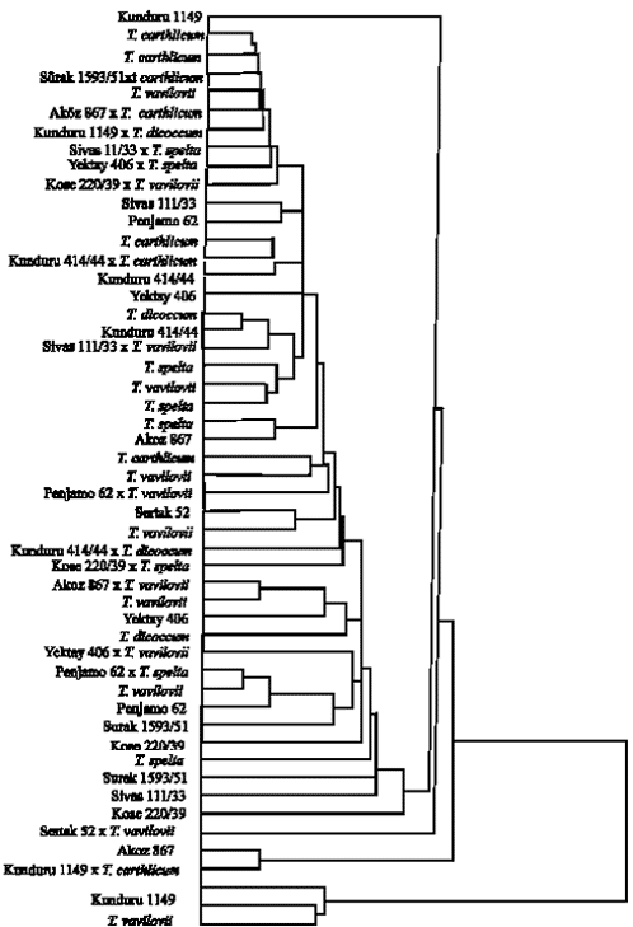
or at a high temperature profile 15/24°C dark/light for a 10/14 h diurnal cycle (Roelfs et al., 1992). Supplementary fluorescent lightning was used to maintain an 18 h day length. In the adult plant resistance for leaf and yellow rusts but due to bad weather conditions and Infection Type (ITs) readings were done according to Stakman and Levine (1962) for the leaf rust resistance as 0 and 1 = resistant, 2 = moderately resistant, 3 = moderately susceptible, 4 = susceptible, X = mixed reaction under the greenhouse conditions. Disease scoring of the plantlets were done for the yellow rusts according toMcNeal et al. (1971) as 0 = immune, 1 = very resistant, 2 = resistant, 3-4 = moderately resistant, 5-6 = moderately susceptible, 7-8 = susceptible and 9 = very susceptible. Obtained all data were analysed by χ2 independency tests and was calculated and the cluster analysis and built the dendrogram (Snedecor and Cochran, 1967).
Electrophoresis: For electrophoresis buffer solution, contains of 1.25 g aluminium lactate, was adjusted to pH 3.1 by lactic acid and 2.0 g saccharose, 0.30 g pyronine and 18.0 mL electrophoresis buffer solution were used for the extract diluting solution. Prepared 100 mL a gel contains 7% (w/v) acrylamide with 0.3% (w/v) bisacrylamide, 24 mg ascorbic acid, 0.4 mg FeSO4.7H2O and 0.25 g aluminium lactate dissolved in 90 mL distilled water and mixture was completed to 100 mL adding electrophoresis buffer solution. In addition and to ensure of polymerization 0.1 mL 3% H2O2 was added, also. Preparing a staining solution, 0.1 g CBB (Commassie Brilliant Blue or R-250) was dissolved into the 95% 10 mL ethylalcohol plus 12% 240 mL TCA (Trichloroacetic acid) in distilled water. For the preparation of destaining solution, 12% TCA was added to distilled water (Zillman and Bushuk, 1979). During this study, Vertical LKB 2001 model cooler and dual tanked electrophoresis apparatus were used to be able to determining hybrid plants, the gliadin banding patterns, one of the seed storage protein, of the various genotypes employed in this study was analyzed by sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE) with some modification and all the procedures were performed according to Bushuk and Zillman (1978). Twelwe-slotted gel plates were run with the first and last end fitted with as a reference band of Canadian wheat cultivar Marquis, which was placed on both sides of the gel. Two parents and all obtained F1 hybrids (each in eight separate slots) were run on the same gel. For maximal separation of gliadin components, electrophoresis timing was followed approximately 2 h and 45` at 10°C under 500 V. After completion of the electrophoresis, each of the gels was transferred to staining tray and applied all related standard procedures described by Zillman and Bushuk (1979). After completing (SDS-PAGE) electrophoresis of the each combination gel plates were photographed (Benedetti et al., 1990). A Canadian wheat cultivar Marquis was used as a reference band for the electrophoregrams of the combinations. It was assigned a relative mobility of 0.50. And other bands were appraised according to the basis of the Marquiss electrophoretic mobility to 0.50 (Bushuk and Zillman, 1978). All combinations` electrophoregams were re-attributed and considered as the regions of a, ß, χ and χ according to molecular weight and band number (Zillman and Bushuk, 1979).
RESULTS AND DISCUSSION
Parents and crosses, genome formulae; scientific name (nomenclature), chromosomal numbers and their responses to the leaf and stripe rusts were presented (Table 1). Genotypes that resistant to rusts and dominance agro-morphological traits were used in a hybridization program as a male parent. In the seedling resistance stage, three main traits (Leaf Colour (LC) (Dark green, Dg and light green, Lg); Auricle Colour (AC) (White (Wh) and Red (Re)) and Leaf Waxiness (LW) (Waxy (Wx) and Non waxy (Nw)) were examined. Due to the theirs dominance inheritance in the seedling resistance stage (Sears, 1974; Skowmand, 1977; Knott, 1987, 1989; Levy and Feldman, 1989; Zhang and Knott, 1990; Kema, 1992; Roelfs et al., 1992; Van-Silfhout, 1993) were selected and examined with the relationship of the leaf and yellow and rusts.
Kunduru 1149 x T. carthlicum
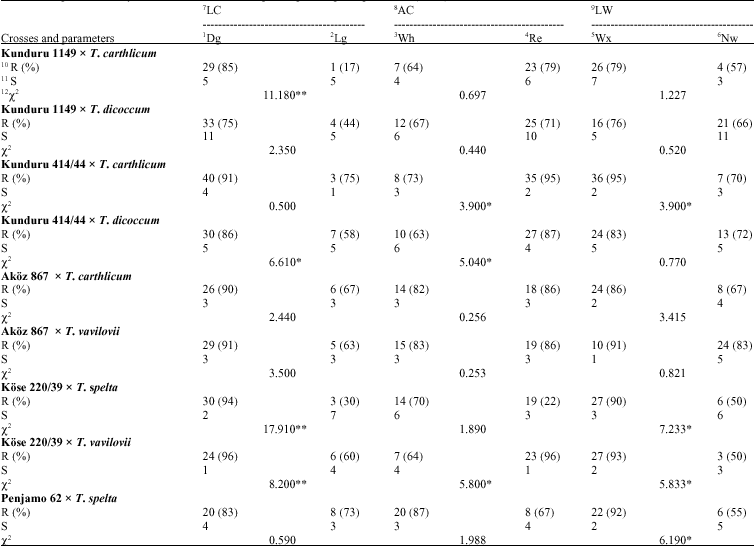
Leaf rust resistance: For the LC; 40 Dg plants were found to be resistant and 1 Lg plant was obtained to be susceptible (χ2 = 12.680, p<0.01) and resistant plant ratios were calculated as 98 and 33%, relatively; for the AC; 11 Wh plants were found to be resistant and 30 Re plant determined was found to be susceptible (χ2 = 0.340, p<0.05) and resistant plant ratios were calculated as 92 and 94%, relatively; for the LW; 36 Wx leaved plants were found to be resistant and 5 Nw leaved plants were fixed to be susceptible (χ2 = 2.110, p>0.05 ) and resistant plant ratios were calculated as 92 and 100%, relatively (Table 2).
Yellow rust resistance: Twenty nine Dg plants were found to be resistant and 1 Lg plant was obtained to be susceptible for the LC (χ2 = 11.180, p<0.01) and resistant plant ratios were calculated as 85 and 17%, relatively; for the AC; 7 Wh plants were found to be resistant and 23 Re plants were determined to be susceptible (χ2 = 0.697, p<0.05) and resistant plant ratios were calculated as 64 and 79%, relatively; for the LW; 26 Wx leaved plants were found to be resistant and 4 Nw leaved plants.
| Table 2: | Responses to the leaf rust for some investigated agro-morphological traits |
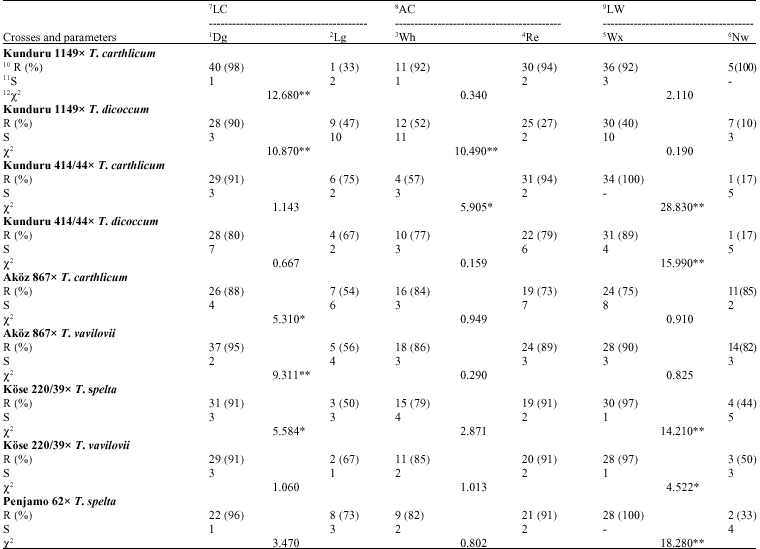 | |
| 1: Dark green, 2: Light green, 3: White, 4: Red, 5: Waxy, 6: Non waxy, 7: Leave colour, 8: Auricle colour, 9: Leaf waxiness, 10: Resistant, 11: Susceptible, 12: Chi-square, **: Statistically significant at the level of 0.1; *: Statistically significant at the level of 0.5 | |
were fixed to be susceptible (χ2 = 1.227, p>0.05) and resistant plant ratios were calculated as 79 and 57%, relatively (Table 3). According to electrophoregram of the combination, gliadin banding pattern distribution was happened as 1, 5, 3 and 6 in a, ß, χ and χ regions, relatively (Fig. 1). It was revealed that 72, 90 and 96 numbered bands were only belong to male parent with the F1 (Fig. 2, 3). These biochemical findings were also verified that this cross is real and it carries the resistance gene(s) of the male parent (Fig. 4).
Kunduru 1149 x T. dicoccum
Leaf rust resistance: For the LC; 28 Dg plants were found to be resistant and 9 Lg plants were obtained to be susceptible (χ2 = 10.870, p<0.01) and resistant plant ratios were calculated as 90 and 40%, relatively; for the AC; 12 Wh plants were found to be resistant and 25 Re plants were determined to be susceptible (χ2 = 10.490, p<0.01) and resistant plant ratios were calculated as 52 and 93%, relatively; for the LW; 30 Wx leaved plants were found to be esistant and 7 Nw leaved plants were fixed to be susceptible (χ2 = 0.190, p>0.05) and resistant plant ratios were calculated as 75 and 70%, relatively (Table 2).
Yellow rust resistance: For the LC; 33 Dg colour leaved plants were found to be resistant and 4 Lg plants were obtained to be susceptible (χ2 = 2.350, p<0.05) and resistant plant ratios were calculated as 75 and 44%, relatively; for the AC; 12 Wh plants were found to be resistant and 25 Re was determined to be susceptible (χ2 = 0.440, p<0.05) and resistant plant ratios were calculated as 67 and 71%, relatively; for the LW; 16 Wx leaved plants were found to be resistant and 21 Nw leaved plants were fixed to be susceptible (χ2 = 0.520, p>0.05) and resistant plant ratios were calculated as 76 and 66%, relatively (Table 3). According to electrophoregram of the combination, gliadin banding pattern distribution was happened as 1, 1, 4 and 9 in a, ß, χ and χ regions, relatively (Fig. 1). It was revealed that 29 and 53 numbered bands were belong to male parent with the F1 (Fig. 2, 3). These biochemical findings were also verified and supported that this cross is real and it carries the resistance gene(s) of the male parent (Fig. 4).
| Table 3: | Responses to the leaf rust for some investigated agro-morphological traits (Cont.) |
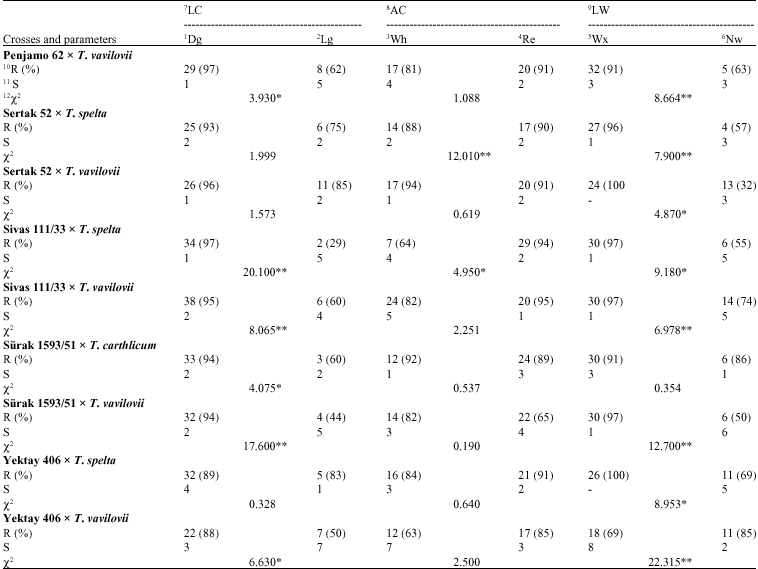 | |
| 1: Dark green, 2: Light green, 3: White, 4: Red, 5: Waxy, 6: Non waxy, 7: Leave colour, 8: Auricle colour, 9: Leaf waxiness, 10: Resistant, 11: Susceptible, 12: Chi-square, **: Statistically significant at the level of 0.1; *: Statistically significant at the level of 0.5 | |
Kunduru 414/44 x T. carthlicum
Leaf rust resistance: Twenty eight Dg plants were found to be resistant and 4 Lg plants were obtained to be susceptible For the LC (χ2 = 0.687, p<0.05) and resistant plant ratios were calculated as 80% and 67%, relatively; for the AC; 10 Wh plants were found to be resistant and 22 Re plants were determined to be susceptible (χ2 = 0.159, p<0.05) and resistant plant ratios were calculated as 77 and 79%, relatively; for the LW; 31 Wx leaved plants were found to be resistant and 1 Nw leaved plant was fixed to be susceptible (χ2 = 15.990, p>0.01) and resistant plant ratios were calculated as 89 and 17%, relatively (Table 2).
Yellow rust resistance: In F2-generation of this cross and for the LC; 30 Dg plants were found to be resistant and 7 Lg plants were obtained to be susceptible (χ2 = 6.610, p<0.01) and resistant plant ratios were calculated as 86% and 58%, relatively; for the AC; 10 Wh plants were found to be resistant and 27 Re plants were determined to be susceptible (χ2 = 5.040, p<0.01) and resistant plant ratios were calculated as 63 and 87%, relatively; for the LW; 24 Wx leaved plants were found to be resistant and 13 Nw leaved plants were fixed to be susceptible (χ2 = 0.770, p>0.05) and resistant plant ratios were calculated as 83 and 72%, relatively (Table 3). According to electrophoregram of this combination, gliadin banding pattern distribution was happened as 2, 2, 2 and 8 in a, ß, χ and χ regions, relatively (Fig. 1). It was revealed that 30 numbered band was belongs to male parent at the same time with the F1 (Fig. 2, 3). These biochemical findings were also verified and supported that this cross is real and carries the resistance gene(s) of male parent (Fig. 4).
Kunduru 414/44 x T. dicoccum
Leaf rust resistance: Twenty nine Dg plants were found to be resistant and 6 Lg plants were obtained to be susceptible (χ2 = 1.143, p<0.05) and resistant plant ratios were calculated as 91 and 75%, relatively; for the AC; 4 Wh plants were found to be resistant and 31 Re plant determined to be susceptible (χ2 = 5.905, p<0.05) and
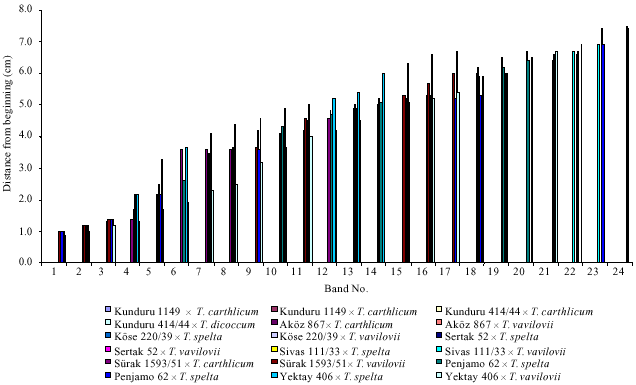 | |
| Fig 1: | Band numbers of the combinations according to the distance from the beginning (cm) |
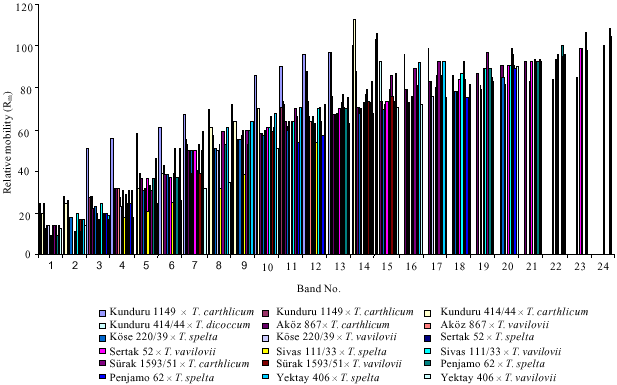 | |
| Fig 2: | Band numbers of the combinations according to the relative mobility (Rm) |
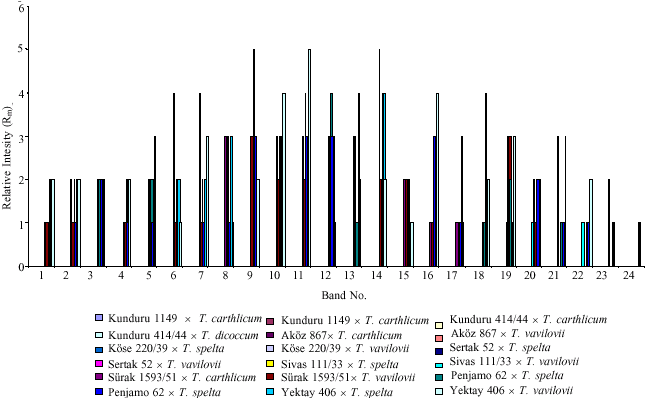 | |
| Fig 3: | Band numbers of the combinations according to relative intensity (Ri) |
resistant plant ratios were calculated as 57 and 94%, relatively; for the LW; 34 Wx leaved plants were found to be resistant and 1 Nw leaved plant was fixed to be susceptible (χ2 = 28.830, p>0.01) and resistant plant ratios were calculated as 34 and 6%, relatively (Table 2).
Yellow rust resistance: In F2-generation of this cross and for the LC; 40 Dg plants were found to be resistant and 3 Lg plants were obtained to be susceptible (χ2 = 0.500, p<0.05) and resistant plant ratios were calculated as 91 and 75%, relatively; for the AC; 8 Wh plants were found to be resistant and 35 Re plants were determined to be susceptible (χ2 = 3.900, p<0.01) and resistant plant ratios were calculated as 73 and 95%, relatively; for the LW; 36 Wx leaved plants were found to be resistant and 7 Nw leaved plants were fixed to be susceptible (χ2 = 3.900, p>0.01) and resistant plant ratios were calculated as 95 and 70%, relatively (Table 3). According to electrophoregram of this combination, gliadin banding pattern distribution was happened as 4, 1, 4 and 8 in a, ß, χ and χ regions, relatively (Fig. 1). It was revealed that 32, 39 and 99 numbered bands were belong to male parent at the same time with the F1 (Fig. 2, 3). These biochemical findings were also verified and supported that this cross is real and carries the resistance gene(s) of male parent (Fig. 4).
AkOz 876 x T. carthlicum
Leaf rust resistance: In F2-generation of this cross and for the LC; 28 Dg plants were found to be resistant and 7 Lg plants were obtained to be susceptible (χ2 = 5.310, p<0.01) and resistant plant ratios were calculated as 88 and 54%, relatively; for the AC; 16 Wh plants were found to be resistant and 19 Re plants were determined to be susceptible (χ2 = 0.949, p<0.05) and resistant plant ratios were calculated as 84 and 73%, relatively; for the LW; 24 Wx leaved plants were found to be resistant and 11 Nw leaved plant was fixed to be susceptible (χ2 = 0.901, p>0.05) and resistant plant ratios were calculated as 75 and 85%, relatively (Table 2). Yellow rust resistance: In F2-generation of this cross and for the LC; 26 Dg plants were found to be resistant and 6 Lg plants were obtained to be susceptible (χ2 = 2.440, p>0.05) and resistant plant ratios were calculated as 90 and 67%, relatively; for the AC; 14 Wh plants were found to be resistant and 18 Re plants were determined to be susceptible (χ2 = 0.256, p>0.05) and resistant plant ratios were calculated as 82 and 86%, relatively; for the LW; 24 Wx leaved plants were found to be resistant and 8 Nw leaved plants were fixed to be susceptible (χ2 = 3.415, p>0.05) and resistant plant ratios were calculated as 86 and 67%, relatively (Table 3). According to electrophoregram of this combination, gliadin banding pattern distribution was happened as 3, 4, 4 and 10 in a, ß, χ and χ regions, relatively (Fig. 1). It was revealed that 16, 22, 67, 71 and 83 numbered bands were belong to male parent at the same time with the F1 (Fig. 2, 3). These biochemical evidences were also verified and supported that this cross is real and carries the resistance gene (s) of male parent (Fig. 4).
Akoz 876 xT. vavilovii
Leaf rust resistance: In F2-generation of this cross and for the LC; 37 Dg plants were found to be resistant and 5 Lg plants were obtained to be susceptible (χ2 = 9.311, p<0.01) and resistant plant ratios were calculated as 95 and 56%, relatively; for the AC; 18 Wh plants were found to be resistant and 24 Re plant determined to be susceptible (χ2 = 0.290, p<0.05) and resistant plant ratios were calculated as 86 and 89%, relatively for the LW; 28 Wx leaved plants were found to be resistant and 14 Nw leaved plants were fixed to be susceptible (χ2 = 0.825, p>0.05) and resistant plant ratios were calculated as 90 and 82%, relatively (Table 2).
Yellow rust resistance: In F2-generation of this cross and for the LC; 29 Dg plants were found to be resistant and 5 Lg plants were obtained to be susceptible (χ2 = 3.500, p<0.05) and resistant plant ratios were calculated as 91 and 63%, relatively; for the AC; 15 Wh plants were found to be resistant and 19 Re plants were determined to be susceptible (χ2 = 0.253, p<0.05) and resistant plant ratios were calculated as 83 and 86%, relatively; for the LW; 10 Wx leaved plants were found to be resistant and 24 non-waxy leaved plants were fixed to be susceptible (χ2 = 0.821, p>0.05) and resistant plant ratios were calculated as 91 and 83%, relatively (Table 3). According to electrophoregram of this combination, gliadin banding pattern distribution was happened as 0, 2, 5 and 10 in a, ß, χ and χ regions, relatively (Fig. 1). It was revealed that 32, 38, 51, 62, 74 and 79 numbered bands were belong to male parent at the same time with the F1 (Fig. 2, 3). These biochemical findings were also verified and supported that this cross is real and carries the resistance gene(s) of male parent (Fig. 4).
KOse 220/39 x T. spelta
Leaf rust resistance: In F2-generation of this cross and for the LC; 31 Dg plants were found to be resistant and 3 Lg plants were obtained to be susceptible (χ2 = 5.584, p>0.05) and resistant plant ratios were calculated as 91 and 50%, relatively; for the AC; 15 Wh plants were found to be resistant and 19 Re plants were determined to be susceptible (χ2 = 2.871, p>0.05) and resistant plant ratios were calculated as 79 and 91%, relatively; for the LW; 30 Wx leaved plants were found to be resistant and 4 Nw leaved plants were fixed to be susceptible (χ2 = 14.210, p<0.01) and resistant plant ratios were calculated as 97 and 44%, relatively (Table 2).
Yellow rust resistance: In F2-generation of this cross and for the LC; 30 Dg plants were found to be resistant and 3 Lg plants were obtained to be susceptible (χ2 = 17.910, p>0.01) and resistant plant ratios were calculated as 94 and 30%, relatively; for the AC; 14 Wh plants were found to be resistant and 19 Re plants were determined to be susceptible (χ2 = 1.890, p>0.05) and resistant plant ratios were calculated as 70 and 86%, relatively; for the LW; 27 Wx leaved plants were found to be resistant and 6Nw leaved plants were fixed to be susceptible (χ2 = 7.233, p<0.01) and resistant plant ratios were calculated as 90 and 50%, relatively (Table 3). According to electrophoregram of this combination, gliadin banding pattern distribution was happened as 3, 1, 7 and 9 in a, ß, χ and χ regions, relatively (Fig. 1). It was revealed that 18, 69 and 73 numbered bands were belong to male parent at the same time with the F1 (Fig. 2, 3). These biochemical findings were also verified and supported that these cross is real and carries the resistance gene (s) of male parent (Fig. 4).
KOse 220/39 x T. vavilovii
Leaf rust resistance: In F2-generation of this cross and for the LC; 29 Dg plants were found to be resistant and 3 Lg plants were obtained to be susceptible (χ2 = 1.050, p>0.05) and resistant plant ratios were calculated as 91 and 67%, relatively; for the AC; 11 Wh plants were found to be resistant and 20 Re plants were determined to be susceptible (χ2 = 1.013, p>0.05) and resistant plant ratios were calculated as 85 and 91%, relatively; for the LW; 28 Wx leaved plants were found to be resistant and 3 Nw leaved plants were fixed to be susceptible (χ2 = 4.522, p<0.05) and resistant plant ratios were calculated as 97 and 50%, relatively (Table 2).
Yellow rust resistance: In F2-generation of this cross and for the LC; 24 Dg plants were found to be resistant and 6 Lg plants were obtained to be susceptible (χ2 = 8.200, p<0.01) and resistant plant ratios were calculated as 96 and 60%, relatively; for the AC; 7 Wh plants were found to be resistant and 23 Re plants were determined to be susceptible (χ2 = 5.800, p<0.05) and resistant plant ratios were calculated as 64 and 96%, relatively; for the LW; 27 Wx leaved plants were found to be resistant and 3 Nw leaved plants were fixed to be susceptible (χ2 = 5.833, p<0.05) and resistant plant ratios were calculated as 93 and 50%, relatively (Table 3). According to electrophoregram of this combination, gliadin banding pattern distribution was happened as 3, 3, 6 and 11 in a, ß, χ and χ regions, relatively (Fig. 1). It was revealed that 14, 39, 66, 77 and 83 numbered bands were only belong to male parent at the same time with the F1 (Fig. 2, 3). These evidences were verified and supported that this cross is real and carries the resistance gene(s) of male parent (Fig. 4).
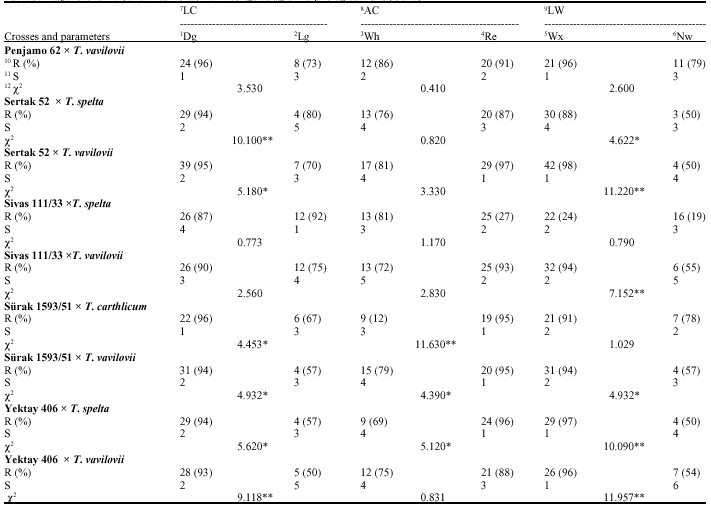
Penjamo 62 x T. spelta
Leaf rust resistance: In F2-generation of this cross and for the LC; 22 Dg plants were found to be resistant and 8 Lg plants were obtained to be susceptible (χ2 = 3.740, p>0.05) and resistant plant ratios were calculated as 96 and 73%, relatively; for the AC; 9 Wh plants were found to be resistant and 21 Re plants were determined to be susceptible (χ2 = 0.802, p>0.05) and resistant plant ratios were calculated as 82 and 91%, relatively; for the LW; 28 Wx leaved plants were found to be resistant and 2 Nw leaved plants were fixed to be susceptible (χ2 = 18.280, p<0.01) and resistant plant ratios were calculated as 100 and 33%, relatively (Table 2).
Yellow rust resistance: In F2-generation of this cross and for the LC; 22 Dg plants were found to be resistant and 6 Lg plants were obtained to be susceptible (χ2 = 4.453, p>0.01) and resistant plant ratios were calculated as 96 and 67%, relatively; for the AC; 9 Wh plants were found to be resistant and 19 Re plants were plant determined to be susceptible (χ2 = 11.630, p>0.05) and resistant plant ratios were calculated as 75 and 95%, relatively; for the LW; 21 Wx leaved plants were found to be resistant and 7 Nw leaved plants were fixed to be susceptible (χ2 = 1.029, p>0.05) and resistant plant ratios were calculated as 91 and 78%, relatively (Table 2). According to electrophoregram of this combination, gliadin banding pattern distribution was happened as 5, 1, 5 and 13 in a, ß, χ and χ regions, relatively. It was revealed that 70, 74, 76, 89 and 100 numbered bands were belong to only male parent at the same time with the F1 (Fig. 2, 3). These biochemical findings were also verified and supported that this cross is real and carries the resistance gene(s) of male parent (Fig. 4).
Penjamo 62 x T. vavilovii
Leaf rust resistance: In F2-generation of this cross and for the LC; 29 Dg plants were found to be resistant and 8 Lg plants were obtained to be susceptible (χ2 = 8.664, p>0.01) and resistant plant ratios were calculated as 97 and 62%, relatively; for the AC; 17 Wh plants were found to be resistant and 20 Re plants were determined to be susceptible (χ2 = 1.088, p>0.05) and resistant plant ratios were calculated as 81 and 91%, relatively; for the LW; 32 Wx leaved plants were found to be resistant and 5 Nw leaved plants were fixed to be susceptible (χ2 = 3.930, p<0.05) and resistant plant ratios were calculated as 91 and 63%, relatively (Table 4).
Yellow rust resistance: In F2-generation of this cross and for the LC; 31 Dg plants were found to be resistant and 4 Lg plants were obtained to be susceptible (χ2 = 4.932,(0.05) and resistant plant ratios were calculated as 94 and 57%, relatively; for the AC; 15 Wh plants were found to be resistant and 20 Re plants were determined to be susceptible (χ2 = 4.390, p>0.05) and resistant plant ratios were calculated as 79 and 95%, relatively; for the LW; 31 Wx leaved plants were found to be resistant and 4 Nw leaved plants were fixed to be susceptible (χ2 = 4.934, p>0.05) and resistant plant ratios were calculated as 94 and 57%, relatively (Table 5). According to electrophoregram of this combination, gliadin banding pattern distribution was happened as 5, 1, 5 and 12 in a, ß, χ and χ regions, relatively. It was revealed that 37, 41, 51, 85 and 98 numbered bands were belong to only male parent at the same time with the F1 (Fig. 2, 3). These biochemical findings were also verified and supported that this cross is real and carries the resistance gene(s) of male parent (Fig. 4).
Sertak 52 x T. speltas
Leaf rust resistance: In F2-generation of this cross and for the LC, 25 dark green plants were found to be resistant and 6 light green plant were obtained to be
| Table 4: | Responses to the yellow rust for some investigated agro-morphological traits (Cont.) |
 | |
| 1: Dark green, 2: Light green, 3: White, 4: Red, 5: Waxy, 6: Non waxy, 7: Leave colour, 8: Auricle colour, 9: Leaf waxiness, 10: Resistant, 11: Susceptible, 12: Chi-square, **: Statistically significant at the level of 0.1; *: Statistically significant at the level of 0.5 | |
susceptible (χ2 = 1.999, p<0.05) and resistant plant ratios were calculated as 97 and 75%, relatively; for the AC;14 Wh plants were found to be resistant and 17 Re plants were determined to be susceptible (χ2 = 12.010, p>0.01) and resistant plant ratios were calculated as 88 and 90%, relatively; for the LW, 27 Wx leaved plants were found to be resistant and 4 Nw leaved plant was fixed to be susceptible (χ2 = 7.900, p<0.01) and resistant plant ratios were calculated as 96 and 57%, relatively (Table 4).
Yellow rust resistance: In F2-generation of this cross and for the LC, 20 Dg plants were found to be resistant and 8 Lg plants were obtained to be susceptible (χ2 = 0.590, p>0.05) and resistant plant ratios were calculated as 94 and 57%, relatively; for the AC; 20 Wh plants were found to be resistant and 8 Re plants were determined to be susceptible (χ2 = 1.988, p>0.05) and resistant plant ratios were calculated as 87 and 67%, relatively; for the LW, 22 Wx leaved plant was found to be resistant and 6 Nw leaved plant was fixed to be susceptible (χ2 = 6.190, p<0.05) and resistant plant ratios were calculated as 92 and 55%, relatively (Table 5). Due to a technical reasons, electrophoresis process of this combination can not be completed and electrophoregrams can not be prepared for the analysis of progeny.
Sertak 52 x T. vavilovii
Leaf rust resistance: In F2-generation of this cross and for the LC, 26 Dg plants were found to be resistant and 11 Lg plants were obtained to be susceptible (χ2 = 1.573, p>0.05) and resistant plant ratios were calculated as 97 and 62%, relatively; for the AC; 17 Wh plants were found to be resistant and 20 Re plants were determined to be susceptible (χ2 = 0.619, p>0.05) and resistant plant ratios were calculated as 94 and 84%, relatively; for the LW; 24 Wx leaved plants were found to be resistant and 13 Nw leaved plants were fixed to be susceptible (χ2 = 4.870, p<0.05) and resistant plant ratios were calculated as 100 and 32%, relatively (Table 4).
Yellow rust resistance: In F2-generation of this cross and for the LC, 24 Dg plants were found to be resistant and 8 Lg plant obtained was to be susceptible (χ2 = 3.530, p>0.05) and resistant plant ratios were calculated as 96
| Table 5: | Responses to the yellow rust for some investigated agro-morphological traits (Cont.) |
 | |
| 1: Dark green, 2: Light green, 3: White, 4: Red, 5: Waxy, 6: Non waxy, 7: Leave colour, 8: Auricle colour, 9: Leaf waxiness, 10: Resistant, 11: Susceptible, 12 : Chi-square, **: Statistically significant at the level of 0.1; *: Statistically significant at the level of 0.5 | |
and 73%, relatively; for the AC; 12 Wh plants were found to be resistant and 20 Re plants were determined to be susceptible (χ2 = 0.410, p>0.05) and resistant plant ratios were calculated as 86 and 91%, relatively; for the LW; 21 Wx leaved plants were found to be resistant and 11 Nw leaved plants were fixed to be susceptible (χ2 = 2.600, p>0.05) and resistant plant ratios were calculated as 96 and 79%, relatively (Table 5). According to electrophoregram of this combination, gliadin banding pattern distribution was happened as 3, 1, 2 and 5 in a, ß, χ and χ regions, relatively (Fig. 1). It was revealed that 70, 74, 76 and 91 numbered bands were only belong to male parent at the same time with the F1 (Fig. 2, 3). These biochemical findings were also verified and supported that this cross is real and carries the resistance gene(s) of male parent (Fig. 4).
Sivas 111/33 x T. spelta
Leaf rust resistance: In F2-generation of this cross and for the LC, 34 Dg plants were found to be resistant and 2 Lg plant obtained was to be susceptible (χ2 = 20.100, p<0.01) and resistant plant ratios were calculated as 97 and 29%, relatively; for the AC; 7 Wh plants were found to be resistant and 29 Re plants were determined to be susceptible (χ2 = 4.950, p>0.05) and resistant plant ratios were calculated as 64 and 94%, relatively; for the LW; 30 Wx leaved plants were found to be resistant and 6 Nw leaved plants were fixed to be susceptible (χ2 = 9.180, p<0.01) and resistant plant ratios were calculated as 97 and 55%, relatively (Table 4).
Yellow rust resistance: In F2-generation of this cross and for the LC, 29 Dg plants were found to be resistant and 4 Lg plants were obtained to be susceptible (χ2 = 10.100, p<0.01) and resistant plant ratios were calculated as 94 and 80%, relatively; for the AC; 13 Wh plants were found to be resistant and 20 Re plants were determined to be susceptible (χ2 = 0.820, p>0.05) and resistant plant ratios were calculated as 76 and 87%, relatively; for the LW; 30 Wx leaved plants were found to be resistant and 6 Nw leaved plants were fixed to be susceptible (χ2 = 4.622, p<0.05) and resistant plant ratios were calculated as 88 and 50%, relatively (Table 5). According to electrophoregram of this combination, gliadin banding pattern distribution was happened as 4, 1, 6 and 2 in a, ß, χ and χ regions, relatively (Fig. 1). It was revealed that 18, 28, 38 and 40 numbered bands were belong to male parent at the same time with the F1 (Fig. 2, 3). These biochemical findings were also verified and supported that this cross is real and carries the resistance gene(s) of male parent (Fig. 4).
Sivas 111/33 x T. vavilovii
Leaf rust resistance: In F2-generation of this cross and for the LC, 38 Dg plants were found to be resistant and 6 Lg plants were obtained to be susceptible (χ2 = 8.065, p<0.01) and resistant plant ratios were calculated as 95 and 60%, relatively; for the AC; 24 Wh plants were found to be resistant and 20 Re plants were determined to be susceptible (χ2 = 2.251, p>0.05) and resistant plant ratios were calculated as 82 and 95%, relatively; for the LW; 30 Wx leaved plants were found to be resistant and 14 Nw leaved plants were fixed to be susceptible (χ2 = 6.978, p<0.01) and resistant plant ratios were calculated as 97 and 74%, relatively (Table 4).
Yellow rust resistance: In F2-generation of this cross and for the LC; 39 Dg plants were found to be resistant and 7 Lg plants were obtained to be susceptible (χ2 = 5.180, p<0.05) and resistant plant ratios were calculated as 95 and 70%, relatively; for the AC; 17 Wh plants were found to be resistant and 29 Re plants were determined to be susceptible (χ2 = 3.330, p>0.05) and resistant plant ratios were calculated as 81 and 97%, relatively; for the LW; 42 Wx leaved plants were found to be resistant and 4 Nw leaved plants were fixed to be susceptible (χ2 = 11.220, p<0.01) and resistant plant ratios were calculated as 98 and 50%, relatively (Table 5). According to electrophoregram of this combination, gliadin banding pattern distribution was happened as 7, 3, 3 and 10 in a, ß, χ and χ regions, relatively (Fig. 1). It was revealed that 14, 20, 30, 56, 64, 77 and 79 numbered bands were belong to only male parent at the same time with the F1 (Fig. 2, 3). These biochemical findings were also verified and supported that this cross is real and carries the resistance gene (s) of male parent (Fig. 4).
Surak 1593/51 x T. carthlicum Leaf rust resistance: In F2-generation of this cross and for the LC; 33 Dg plants were found to be resistant and 3 Lg plants were obtained to be susceptible (χ2 = 4.075, p<0.05) and resistant plant ratios were calculated as 89 and 57%, relatively; for the AC; 12 Wh plants were found to be resistant and 24 Re plants were determined to be susceptible (χ2 = 0.537, p>0.05) and resistant plant ratios were calculated as 92 and 89%, relatively; for the LW; 30 Wx leaved plants were found to be resistant and 6 Nw leaved plants were fixed to be susceptible (χ2 = 0.354, p>0.05) and resistant plant ratios were calculated as 91 and 86%, relatively (Table 4).
Yellow rust resistance: In F2-generation of this cross and for the LC; 26 Dg plants were found to be resistant and 12 Lg plants were obtained to be susceptible (χ2 = 0.773, p>0.05) and resistant plant ratios were calculated as 87 and 92%, relatively; for the AC; 13 Wh plants were found to be resistant and 25 Re plants were determined to be susceptible (χ2 = 1.170, p>0.05) and resistant plant ratios were calculated as 81 and 93%, relatively; for the LW; 22 Wx leaved plants were found to be resistant and 16 Nw leaved plants were fixed to be susceptible (χ2 = 0.790, p>0.05) and resistant plant ratios were calculated as 92 and 84%, relatively (Table 5). According to electrophoregram of this combination, gliadin banding pattern distribution was happened as 5, 3, 5 and 7 in a, ß, χ and χ regions, relatively (Fig. 1). It was revealed that 31, 51, 60, 66, 71, 89, 97 and 99 numbered bands were belong to only male parent at the same time with the F1 (Fig. 2, 3). These biochemical findings were also verified and supported that this cross is real and carries the resistance gene(s) of male parent (Fig. 4).
Surak 1593/51 x T. vavilovii
Leaf rust resistance: In F2-generation of this cross and for the LC; 32 Dg plants were found to be resistant and 4 Lg plants were obtained to be susceptible (χ2 = 17.600, p>0.01) and resistant plant ratios were calculated as 94 and 44%, relatively; for the AC; 14 Wh plants were found to be resistant and 22 Re plants were determined to be susceptible (χ2 = 0.190, p>0.05) and resistant plant ratios were calculated as 82 and 65%, relatively; for the LW; 30 Wx leaved plants were found to be resistant and 6 Nw leaved plants were fixed to be susceptible (χ2 = 12.700, p<0.01) and resistant plant ratios were calculated as 97 and 50%, relatively (Table 4).
Yellow rust resistance: In F2-generation of this cross and for the LC; 26 Dg plants were found to be resistant and 12 Lg plants were obtained to be susceptible (χ2 = 2.650, p>0.05) and resistant plant ratios were calculated as 90 and 75%, relatively; for the AC; 13 Wh plants were found to be resistant and 25 Re plants were determined to be susceptible (χ2 = 2.830, p>0.05) and resistant plant ratios were calculated as 72 and 93%, relatively; for the LW; 32 Wx leaved plants were found to be resistant and 6 Nw leaved plants were fixed to be susceptible (χ2 = 7.152, p<0.01) and resistant plant ratios were calculated as 94 and 55%, relatively (Table 5). According to electrophoregram of this combination, gliadin banding pattern distribution was happened as 2, 3, 5 and 10 in a, ß, χ and χ regions, relatively (Fig. 1). It was revealed that 24, 31, 76 and 93 numbered bands were belong to only male parent at the same time with the F1 (Fig. 2, 3). These biochemical findings were also verified and supported that this cross is real and carries the resistance gene(s) of male parent (Fig. 4).
Yektay 406 x T. spelta
Leaf rust resistance: In F2-generation of this cross and for the LC; 32 Dg plants were found to be resistant and 5 Lg plants were obtained to be susceptible (χ2 = 0.328, p>0.05) and resistant plant ratios were calculated as 89 and 83%, relatively; for the AC; 16 Wh plants were found to be resistant and 21 Re plants were determined to be susceptible (χ2 = 0.640, p>0.05) and resistant plant ratios were calculated as 84 and 91%, relatively; for the LW; 26 Wx leaved plants were found to be resistant and 11 Nw leaved plants were fixed to be susceptible (χ2 = 8.953, p<0.01) and resistant plant ratios were calculated as 100 and 69%, relatively (Table 4).
Yellow rust resistance: In F2-generation of this cross and for the LC; 29 Dg plants were found to be resistant and 4 Lg plants were obtained to be susceptible (χ2 = 5.620, p<0.05) and resistant plant ratios were calculated as 94 and 57%, relatively; for the AC; 9 Wh plants were found to be resistant and 24 Re plants were determined to be susceptible (χ2 = 5.120, p<0.5) and resistant plant ratios were calculated as 69 and 96%, relatively; for the LW; 29 Wx leaved plants were found to be resistant and 4 Nw leaved plants were fixed to be susceptible (χ2 = 10.090, p>0.01) and resistant plant ratios were calculated as 97 and 50%, relatively (Table 5). According to electrophoregram of this combination, gliadin banding pattern distribution was happened as 3, 1, 6 and 7 in a, ß, χ and χ regions, relatively (Fig. 1). It was revealed that 61, 75 and 83 numbered bands were belong to only male parent at the same time with the F1 (Fig 2, 3). These biochemical findings were also verified and supported that this cross is real and carries the resistance gene(s) of male parent (Fig. 4).
Yektay 406 xT. vavilovii
Leaf rust resistance: In F2-generation of this cross and for the LC; 22 Dg plants were found to be resistant and 7 Lg plants were obtained to be susceptible (χ2 = 6.330, p<0.01) and resistant plant ratios were calculated as 88 and 50%, relatively; for the AC; 12 Wh plants were found to be resistant and 17 Re plants were determined to be susceptible (χ2 = 2.500, p>0.05) and resistant plant ratios were calculated as 63 and 85%, relatively; for the LW; 18 Wx leaved plants were found to be resistant and 11 Nw leaved plants were fixed to be susceptible (χ2 = 22.315, p<0.01) and resistant plant ratios were calculated as 69 and 85%, relatively (Table 4).
Yellow rust resistance: In F2-generation of this cross and for the LC; 28 Dg plants were found to be resistant and 5 Lg plants were obtained to be susceptible (χ2 = 0.773, p<0.01) and resistant plant ratios were calculated as 93 and 50%, relatively; for the AC; 12 Wh plants were found to be resistant and 21 Re plants were determined to be susceptible (χ2 = 0.831, p>0.05) and resistant plant ratios were calculated as 75 and 88%, relatively; for the LW; 26 Wx leaved plants were found to be resistant and 7 Nw leaved plants were fixed to be susceptible (χ2 = 11.957, p<0.01) and resistant plant ratios were calculated as 92 and 84%, relatively (Table 5). According to electrophoregram of this combination, gliadin banding pattern distribution was happened as 3, 2, 5 and 12 in a, ß, χ and χ regions, relatively (Fig. 1). It was revealed that 18, 26, 72 and 75 (four bands) numbered bands were belong to only male parent at the same time with the F1 (Fig 2, 3). These biochemical findings were also verified and supported that this cross is real and carries the resistance gene(s) of male parent (Fig. 4).
In terms of the electrophoretical band distribution, relevant bands between the male parents and F1 crosses were varied among (5-7) in durums and (6-14) in aestivums. T. carthlicum was crossed with two durum (Kunduru 1149 and Kunduru 414/44) and aestivums (AkOz 867 and Surak 1593-51) (Fig. 1-3). On the other hand, these bands were found as 61 (1 band) in durums and 51, 71, 86 (3 bands) were fixed in aestivums. T. dicoccum was only crossed with two durum cultivars (Kunduru 1149 and Kunduru 414/44). Two bands, (53 and 61) was shared between them. Seven crosses were formed with the male parent of T. vavilovii. According to its electrophoregram, it was obtained crosses were shared separately with twenty one related bands (14, 17, 20, 31, 32, 37, 51, 53, 57, 58, 62, 70, 71, 74, 76, 83, 89, 93, 94, 96, 99). Similarly, T. spelta, one of the male parents, 21 bands were also common (Fig. 4). In addition, electrophoretic banding pattern was observed in crosses and all associated bands were distributed separately. 14, 51, 64, 73 numbered bands were recorded as shared and they were found as 61 in durums and 51, 71, 86 in aestivums (Fig. 2-4). It can be explained that male parents and derived F1 crosses have the same genetic structure. All obtained data were verified that resistances of the F1 crosses. Again, males were successfully transmitted resistance genes to the F1 crosses that approved by related seedling resistance tests again to examined by leaf and yellow rusts (Fig. 5). Beyond of these, common and durum genotypes have had the same electrophoretical bands and this situation was observed much more in aestivums than durums (up to 14). It can be consider that cytoplasmic interactions, environmental conditions, used equipments and following procedures were made major effect. Separated bands were runned and mostly accumulated at the (χ) regions-more
 | |
| Fig. 5: | Seeding responses in the crosses after inoculations of the leaf (above) and yellow (below) rusts |
than 100 Rm, due to the (very) high relative band densities (Fig. 3). This phenomenon was observed much more in aestivums. And, it can be explain as male parents` possible ploidy. Doubtless, this effect of ploidy seems to be a dilution effect, but no chemical basis for a change in resistance as a result of a change in ploidy level is known (Roelfs et al., 1992) (especially in the crosses of Penjamo 62, AkOz 867 and KOse 220/39). Similar situation was observed very clearly when T. carthlicum, T. spelta and T. vavilovii used as male parents in the study. But, in terms of electrophoretic band separation and in number of common band with male and F1 crosses can be mentioned that these male parents were shown their banding activities reversely (Fig. 1-3). However, the mechanisms of the phenomenon are not clear and our data do not provide a conclusive explanation.
CONCLUSIONS
The first step in identifying a new source of resistance is to test it with a range of relevant pathotypes of the pathogene(s) of interest (Bariana and McIntosh, 1993). Due to the low frequency of resistant plants in a cross and the possibility that they may be overwhelmed by heavy spore loads, breeders may have to concentrate on larger populations of fewer crosses. Some limited selection can be done concentrated in families in later generations and selection for virulence by growing hosts with race-specific resistance plays a major role in determining the race structure of the pathogen (Knott and Bai, 1994). Selections have been made on the progenies of the crosses, in order to identify lines which combine disease resistance with valuable agronomic traits. For transferring rust resistance to the wheat from its relatives, a search for new genes should be first made in wheat progenitors having one or more genome in common wheats with (Dg) coloured leaves, (Re) coloured auricles and (Wx) leaves in the seedling resistance were found as dominant characters as described by Dhaliwal et al. (1993) and all evaluations were done with compare of parental traits. Own these characters` relationship with leaf and yellow rusts, it can be easily make pre-selection as a marker trait especially in the number of limited or valuable plants. These results shows and supported that the resistance to leaf rust is dominant and governs by one gene and this outcome supported by Nanda et al. (1983).
REFERENCES
- Bariana, H.S. and R.A. McIntosh, 1993. Cytogenetic studies in wheat. XV. Location of rust resistance genes to VPM1 and their genetic linkage with other disease resistance genes in chromosome 2A. Genome, 36: 476-482.
Direct Link - Benedetti, S., M. Chiaffi, C. Tomassini, D. Lafiandra and E. Porceddu, 1990. One-Dimensional Electrophoretic Separation of Gliadins in a Durum Wheat Collection from Ethiopia. In: Wheat Genetic Resources: Meeting Diverse Needs, Srivastava, J.P. and A.B. Damania (Eds.). John Wiley and Sons, Inc, New York, USA., pp: 89-100.
- Bushuk, W. and R.R. Zillman, 1978. Wheat cultivar identification by gliadin electrophoregrams. I. Apparatus, method and nomenclature. Can. J. Plant. Sci., 58: 505-515.
Direct Link - Dhaliwal, H.S., H. Singh, S. Gupta, P.S. Bagga and K.S. Gill, 1991. Evaluation of aegilops and wild Triticum species for resistance to leaf rust (Puccinia recondita f. sp. tritici) of wheat. Int. J. Trop. Agric., 9: 118-122.
Direct Link - Feldman, M. and E.R. Sears, 1981. The wild gene resources of wheat. Sci. Am., 244: 102-112.
Direct Link - Flor, H.H., 1956. The complementary genic systems in flax and flax rust. Adv. Genet., 8: 29-54.
CrossRefDirect Link - Kema, G.H.J., 1992. Resistance in spelt wheat to yellow rust. I. Formal analysis and variation for gliadin patterns. Euphytica, 63: 225-231.
CrossRefDirect Link - Knott, D.R., 1987. Transferring Alien Genes to Wheat. In: Wheat and Wheat Improvement, Heyne, E.G. (Ed.). 2nd Edn., American Society of Agronomy. Madison,Wisconsin, USA., pp: 462-471.
Direct Link - Knott, D.R., 1989. The effect of transfers of alien genes for leaf rust resistance on the agronomic and quality characteristics of wheat. Euphytica, 44: 65-72.
CrossRefDirect Link - Knott, D.R. and D. Bai, 1994. Genetic studies of leaf and stem rust resistance in six accessions of Triticum turgidum var. dicoccoides. Genome, 37: 405-409.
Direct Link - Levy, A.A. and M. Feldman, 1988. Ecogeographical distribution of HMW-glutenin alleles in populations of the wild tetraploid wheat Triticum turgidum var. dicoccoides. Theor. Applied Genet., 75: 651-658.
CrossRefDirect Link - Nanda, G.S., A. Nityagopal and K.S. Gil, 1983. Genetic analysis of yellow and brown rust interaction with certain aspects of physiology, quality and yield components in Triticum aestivum L. Proceedings of the 6th International Wheat Genetics Symposium, November 28-December 3, 1983, Kyoto, Japan, pp: 291-296.
- Ozgen, M., M. Yildiz, H. Ulukan and N. Koyuncu, 2004. Association of gliadin protein pattern and rust resistance derived from Aegilops umbellulata Zhuk. in winter Triticum durum Desf. Desf. Breed. Sci., 54: 287-290.
CrossRefDirect Link - Roelfs, A.P., R.P. Singh and E.E. Sari, 1992. Rust Diseases of Wheat: Concepts and Methods of Disease Management. CIMMIYT, Mexico, ISBN: 9789686127478, Pages: 81.
Direct Link - Zadoks, J.C., T.T. Chang and C.F. Konzak, 1974. A decimal code for the growth stages of cereals. Weed Res., 14: 415-421.
CrossRefDirect Link - Zhang, H.M. and D.R. Knott, 1990. Inheritance of leaf rust resistance in durum wheat. Crop Sci., 30: 1218-1222.
Direct Link - Zillman, R.R. and W. Bushuk, 1979. A wheat cultivar identification by gliadin electrophoregrams. II. Effects of environmental and experimental factors on the gliadin electrophoregrams. Can. J. Plant Sci., 59: 281-286.
Direct Link