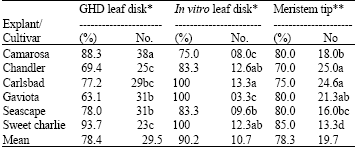
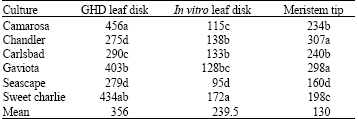
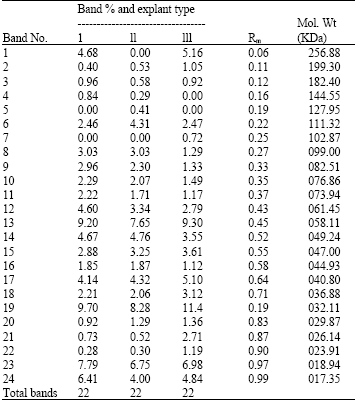
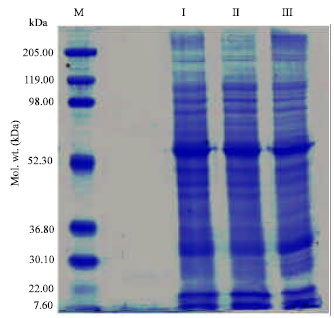
Research Article
High Frequency, Direct Shoot Regeneration from Greenhouse-Derived Leaf Disks of Six Strawberry Cultivars
Department of Horticulture, Faculty of Agriculture, Suez Canal University Ismailia, Egypt
Mohamed S. Beltagi
Department of Botany, Faculty of Science, Suez Canal University Ismailia, Egypt
Mona A. Ismail
Department of Botany, Faculty of Science, Suez Canal University Ismailia, Egypt
Ginesia F. Omar
Department of Horticulture, Faculty of Agriculture, Suez Canal University Ismailia, Egypt