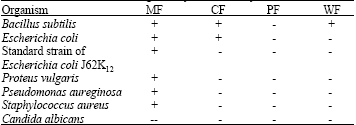
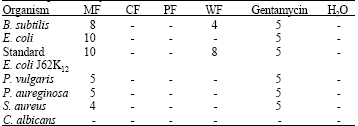
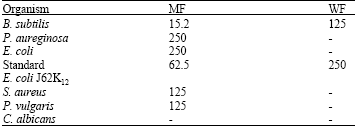
Research Article
Preliminary Investigation on the Phytochemistry and Antimicrobial Activity of Senna alata L. Flower
Department of Botany
S.E. Omonigho
Department of Microbiology,University of Benin, PMB 1154, Benin City, Nigeria
C.L. Igeleke
Department of Basic and Applied Sciences, Benson Idahosa University, Benin City, Nigeria