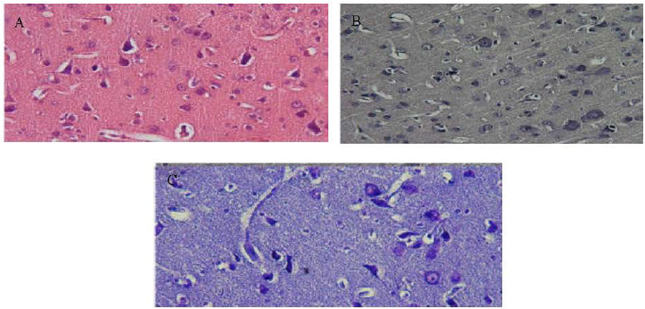
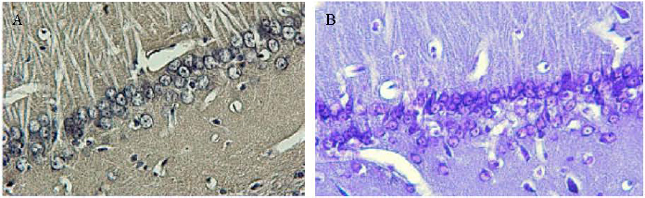
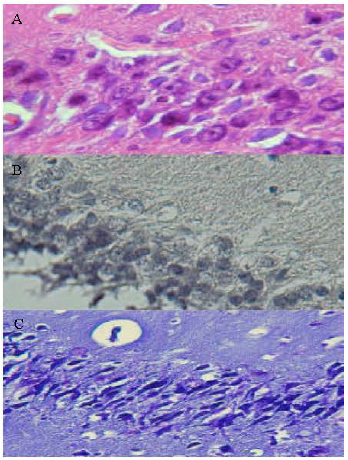
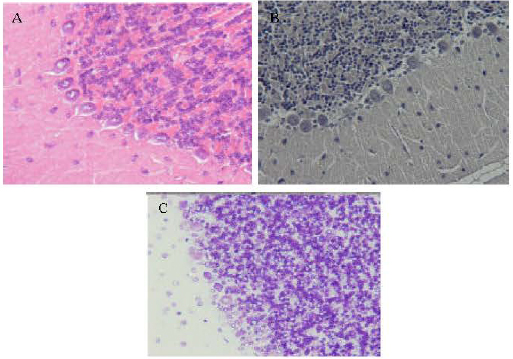
Research Article
Effects Modification of Iron Hematoxylin on Neuron Staining
Department of Anatomy, Gorgan University of Medical Sciences Gorgan, Iran
M.J. Golalipour
Department of Anatomy, Gorgan University of Medical Sciences Gorgan, Iran
R. Ghorbani
Reproductive Research Center, Kerman Shah University of Medical Sciences, Kerman Shah, Iran
M. Khazaei
Reproductive Research Center, Kerman Shah University of Medical Sciences, Kerman Shah, Iran