ABSTRACT
This study was undertaken to examine the unresolved questions surrounding the influence of silicon softener on cotton fabrics. Results showed that the synthesized silicon softener was comparable with other tested conventional softener, According to present investigations the emulsions E1 and E2 is not economical and it is not evenly qualified and it become two phased after 24 h. But emulsion E3 was even and more economical. Moreover, it has high stability. In addition, measurement of kinetic and static friction show that the general effect of silicon softener on cotton cloth is the decrease of friction. Also, it was concluded that with increasing the add on percentage of softener, the crease of reflection angle did not change bending length and static and kinetic friction index significantly.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2007.676.678
URL: https://scialert.net/abstract/?doi=pjbs.2007.676.678
INTRODUCTION
One of the most important factor in fabrics is it’s softness (Koc and Zervent 2006; Edges, 1998). According to the losing of wax/ oil during wet finished process and washing (Cote, 2002). The friction between fibers increase and due to a harsh handle (Funder et al., 1997; Mehra et al., 1991). For solving the mentioned problem applying of softeners have been conventional. Between different kinds of softener, two main categories have been more going: Cationic and silicon softener (Mooney, 1980). This kind of softener according to stability is one of permanent softeners and keeps the softness even often several times of washing (Salvi, 1980). In an investigations carried by Lee and Kim (2005), cotton fabrics were treated with a glyoxal/glycol mixture for a nonformaldehyde durable- press finish by a pad-dry-cure method. At this way the effects of additives such as sodium hydrogen sulfate, polyurethane and a silicon softener were examined. Giolando et al. (1994), have introduced the environmental safety database for a new fabric softening cationic surfactant. Furthermore, Roghair et al. (1992), Leeuwen et al. (2003), have examined the toxicological risk evaluation of the cationic fabrics softener" Ditallow dimethyl ammonium Chloride (DTDMAC)". An et al. (2006) at similar experiments, have examined the characterization and file morphology of dodecylpolysiloxane. In this study it was intended to synthesis a silicon softener in an uncomplicated method of a silicon based oil, to compare the synthesized softener with some conventional softeners used in industry presently and to investigate the dependence of parameters: CRS and binding length and static and kinetic friction index on the concentration of silicon softener.
MATERIALS AND METHODS
Silicon oil is unsolvable in water, for solving it in water and forming an emulsion used suitable emulsifier by, nonionic soap with ethylene oxide mol 6 (nonionic surfactant 100%), Texapon N70 and mono ethylene glycol. Table 1 shows the percent of ingredients in 3 kind emulsifiers (Howell and Mazurji, 1959).
This investigation was carried out on April, 2005 at Textile engineering department of Eslamsheahr Azad university and Sayar company. At the beginning, 3 emulsifiers (E1, E2 and E3) are mixed with silicon oil. Table 2 shows the optimized concentration for each of emulsifier. The samples prepared at 30-40°C.
In this experiment the physical quality of fabric is measured by using the different viscosity of the silicon softener (Dreby, 1942). The samples prepared at 5, 12, 25, 55 and 100 g L-1 concentration. For having suitable pH in the bath, 1-2 g L-1 acid acetic was used and amount of add-on is 1-2% (Table 3).
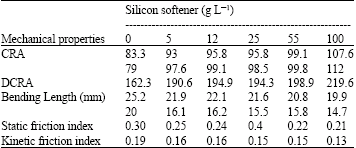
| Table 1: | Result of mechanical properties fabric sample with softener |
 | |
| Table 2: | Percent of ingredients in three kinds of emulsifiers |
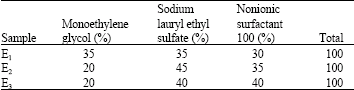 | |
| Table 3: | Amount of ratio (softener/add-on) |
 | |
| Table 4: | Result of mechanical properties fabric sample with softener |
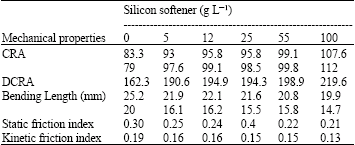 | |
After preparing each bath the sample fabric stays in for a minute. Then, it is padding and dried for 2 and a half minutes in oven. The prepared samples are put in standard conditions of 65% relatively moisture and 20 + 2°C for 48 h. Specimens were examined for their mechanical properties. The results are shown in Table 4.
Figure 1 shows the relationship between CRA (Crease of reflection angle) and silicon-based softener add- on (%). By using 12 g L-1 softener CRA Completely increased. Whereas using up to 50 g L-1 does not have any effect on the increase of CRA and the changes are slow.
DCRA = CRA(warp) + CRA(weft) |
Figure 2 shows the relationship between silicone- based softener add- on(%) and bending length.
Kinetic and static friction index of the samples have been measured and the result is shown in Fig. 3.
The result was investigated with FT- IR spectroscopy. In Fig. 4 shows IR spectroscopy of silicon softener.
RESULTS AND DISCUSSION
Mechanical properties: In order to compare the physical quality of fabrics, we have used five different viscosities of the silicon softener (5, 12, 25, 55 and 100 g-1, respectively). The mechanical properties fabric sample with softener, indicated that all property index including: CRA (Crease of reflection angle), DCRA, bending length, static friction and kinetic friction are shown in Table 1. The results indicated the amounts of the properties will change from 8.3-107.6, 162.3-219.6, 25.2-19.9, 0.30-0.21 and 0.19-0.13, respectively, when the softener concentration increased from 0 to 100 g L-1.
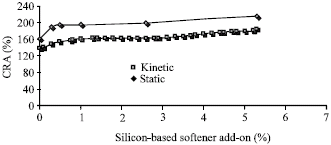 | |
| Fig. 1: | Effect of silicon softener on CRA |
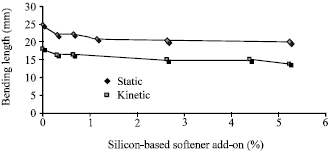 | |
| Fig. 2: | Effect of silicon softener on bending length |
Figure 1 also has certified the results. At this way, CRA will increased by decreasing softener concentration.
Figure 2 shows effect of silicon softener on bending length, the bending length of the showed significant decrease, comparing with reference sample. Additionally, according to results obtained from Table 2, the emulsions E1 and E2 is not economical and it is not evenly qualified and it become two phased after 24 h. But emulsion E3 was even and more economical. Moreover, it has high stability. At Fig. 3 we have examined the effects of silicon softener on static and kinetic friction index on a cotton cloth, measuring kinetic and static friction showed that the friction will decreased highly. The results were certified by FT-IR spectroscopy (Fig. 4). FT-IR spectroscopy of silicon softener in Fig. 4 shows 3 peaks:
| • | Si-O, 1200 cm-1 |
| • | Si-Me, 700 cm-1 |
| • | OH (ethylene glycol), 3100-3600 cm-1 |
These results are in agreement by which was reported sooner by Thorndike and Varley (1961), Wilson (1963), Ohsawa and Namiki (1966) and Ajayi (1992). In order to compare percentage of ingredients at three kinds of emulsifiers, we have mixed Monoethylene glycol, Sodium lauryl ethyl sulfate and non- ionic surfactant 100% at different concentrations (Table 2).
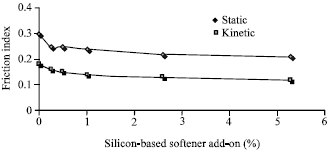 | |
| Fig. 3: | Effect of silicon softener on static and kinetic friction index |
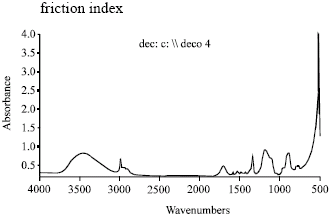 | |
| Fig. 4: | FT- IR spectroscopy of silicon softener |
CONCLUSIONS
To conclude, using of Softeners will decrease the friction between fabrics. Silicon softener also decreases kinetic and static friction of the cloth. By increasing density of the softener, friction decreases. Kinetic friction index is always lower than static friction index. The effect of this softener on cotton fabric is the increase of CRA. These changes are visible even in the least amount of softener. This softener causes the slip of filaments over each other and as a result CRA increases.
The decrease of bending length is also recognizable in warp and wool. Softener causes increasing filaments slipping. The advantages of this softener:
| • | It is permanent and doesn’t vanish by washing several times. |
| • | It also decreases the friction of cloth and makes it soft. |
| • | These groups of softeners are adaptable with various colors and completing materials, so they are usable during dyeing or completion processes. |
| • | These groups of softeners easily changed to emulsion in cold water and give the fabric special underlying. |
REFERENCES
- An, Q., C. Guangwen and L. Linsheng, 2006. Synthesis, characterization and film morphology of deodecylpolysiloxane. J. Applied Polymer Sci., 101: 4480-4486.
Direct Link - Giolando, S.T., R.A. Rapaport, R.J. Larson, T.W. Federlem and P. Masscheylen, 1995. Environmental fate and effects of DEEDMAC: A new rapidly biodegradable cationic surfactant for use in fabric softeners. Chemosphere, 30: 1067-1083.
Direct Link - Koc, E. and B. Zervent, 2006. An experimental approach on the performance of towels-PartI. Bending resistance or softness analysis. Fibers Textiles Eastern Eur., 4: 39-45.
Direct Link - Lee, E.S. and S. Kim, 2005. Effect of additives on durable-press cotton fabrics treated with a glyoxal/glycol mixture. J. Applied Polymer Sci 96: 975-978.
Direct Link - Leeuwen, K.V., C. Roghair, T. Nijs and J. Greef, 2003. Ecotoxicological risk evaluation of the cationic fabric softener DTDMAC. III. Risk assessment. Chemosphere, 24: 629-639.
Direct Link - Roghair, C.J., A.B. Buijze and H.N.P. Schoon, 1992. Ecotoxicological risk evaluation of the cationic fabric softener DTDMAC. I. Ecotoxicological effects. Chemosphere, 24: 599-609.
Direct Link








