ABSTRACT
A protocol has been developed for plant regeneration from protoplast culture of Crocus pallasii subsp. haussknechtii using regenerable embryogenic calli obtained from shoot meristem culture on MS+9.28 μM kinetin+4.52 μM 2,4-D. Protoplasts were isolated directly from embryogenic calli, embedded in Ca-alginate beads and cultured with nurse cells in MS+4.64 μM kinetin+4.52 μM 2,4-D+5.68 μM ascorbic acid+0.3 M mannitol at 20±2°C in darkness. After appearing of microcalli on the surface of the beads, they were transferred onto 1/2MS+2.32 μM kinetin+2.26 μM 2,4-D+5.68 μM ascorbic acid for growth of embryogenic calli. Somatic embryos matured on MS medium growth regulator free and germinated on 1/2MS+14.45 μM GA3 +4.43 μM BA at 20±2°C in a 16/8 h light/dark cycle.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2007.659.663
URL: https://scialert.net/abstract/?doi=pjbs.2007.659.663
INTRODUCTION
The culture and regeneration of protoplasts are important steps in somatic hybridization and realization of genetic manipulation of economically valuable plants. However, for the successful application of these techniques the availability of efficient procedures for isolation, cultivation and regeneration are prerequisites (Ma et al., 2003). A frequent problem is that plant protoplasts many require nurse cultures or plating at high densities (often many thousands of cells mL-1) for survival and growth. Additionally, in monocots including Crocus, culture of protoplasts isolated directly from the plant is difficult (Vasil, 1983; Novak, 1990; Vasil and Vasil, 1992) and protoplasts derived from cell-suspension often have failed to produce green plants (Maddock, 1987; Lee et al., 1988). Indeed, plant regeneration in this genus has been usually successful when embryogenic calli were used as the source of protoplasts (Vasil, 1988; Isa et al., 1990; Karamian and Ebrahimzadeh, 2001). Protoplast culture in Crocus sativus hasbeen attempted through combination of alginate entrapment and nurse culture method earlier (Isa et al., 1990; Ebrahimzadeh et al., 2000a). In recent years, plantlet regeneration from protoplast derived embryogenic calli of Crocus cancellatus has been reported (Karamian and Ebrahimzadeh, 2001). The present study reports for the first time the isolation and culture of protoplasts directly from embryogenic calli derived from shoot meristem culture of C. pallasii subsp. haussknechtii without going through a cell-suspension system and subsequent plant regeneration.
MATERIALS AND METHODS
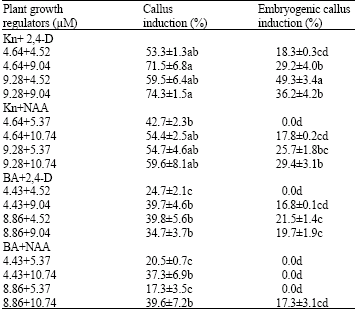
Induction of embryogenic callus: Crocus pallasii subsp. haussknechtii corms were collected from Dalakhani mountain near Kermanshah province of Iran during two annual flowering seasons, in October 2004 and 2005. Sprouted corms were utilized as source of explants. Bulblets were separated and washed with tap water and surface sterilized in 0.15% HgCl2 solution for 10 min followed by rinsing 3 times with sterile distilled water. Sterilized shoot meristems were dissected and served as explants. The basal medium consisted of MS mineral salts and organic nutrients (Murashige and Skoog, 1962), 3% sucrose and 0.8% agar. The basal medium was fortified with different concentrations of BA, Kinetin, NAA and 2,4-D as listed in Table 1. The medium pH was adjusted to 5.75 before autoclaving at 121°C and 104 kPa. The cultures were incubated in dark at 20±2°C. Twenty explants per treatment with three replicates were tested. The data for callus initiation were scored after 6 weeks of culture. Callus and embryogenic callus induction frequencies were calculated as the percentage of cultured shoot tips producing callus and embryogenic callus respectively. The data means from all replications were statistically analyzed using a SAS (1987) and separated by DMRT.
Selection and maintenance of embryogenic callus: After 6 weeks of culture, calli were produced from shoot meristems. Nonembryogenic calli were soft and translucent while embryogenic calli were compact and granular. Embryogenic nature of cultures was maintained by visual identification and selection of embryogenic sectors and removal of soft and translucent nonembryogenic portions at the time of subculturing.
| Table 1: | Morphogenetic response of shoot meristem culture of C. pallasii subsp. haussknechtii* |
 | |
*Percentage of explants inducing callus was evaluated after 6 weeks of cultures. Value is the mean±SE of three replicates. Data within a column followed by different letter(s) differ significantly from each other at p≤0.05 | |
For maintenance of embryogenic potential, embryogenic calli together with globular embryos were transferred to MS medium supplemented with 9.28 μM kinetin, 4.52 μM 2,4-D, 5.68 μM ascorbic acid and 3% sucrose. The cultures were incubated at 20±2°C in darkness, subcultured at 21-28 day intervals for 2-3 months and then used for protoplast isolation.
Isolation and culture of protoplasts: Embryogenic calli subcultured for 2-3 months were transferred to a fresh maintenance MS medium for 1-2 weeks. The calli were incubated in a filter-sterilized enzyme solution consisting of MS medium with 0.2% (w/v) Pectolyase Y23 (Sigma), 1% Cellulase Onozuka RS (Sigma), 1% Deriselase, 0.1% MES (2-N-morpholinoethane sulfonic acid) and 0.3 M mannitol at pH 5.7-5.8. The mixture was placed on a rotary shaker (90 rpm) for 1 h and then kept in a stationary position for another 1 h at 25±2°C in darkness. The incubation mixture was then filtered through a 60 μm nylon mesh sieve and washed twice with a washing solution (0.1% MES in 0.3 M mannitol at pH 5.8) to purify the isolated protoplasts. Then, they were layered onto 20% sucrose and centrifuged for 5 min at 100 g. Viable and intact protoplasts floating at the interface were removed with a sterile pasteur pipette and washed twice with protoplast culture medium. The purified protoplasts were mixed gently with 2% sterile Na-alginate solution (Riedel-deHaën) in 0.3 M mannitol at the density of 1x106 protoplasts mL-1. These alginate solutions with the protoplasts were added drop by drop with a sterile pasteur pipette into MS medium containing 1% CaCl2 and 0.3 M mannitol. Each droplet immediately formed an alginate bead. The beads were left in this solution for 20 min to complete gelation, then washed twice with MS medium containing 4.46 μM kinetin, 4.52 μM 2,4-D, 5.68 μM ascorbic acid and 0.3 M mannitol. Cultures were incubated in the same medium. For nurse cultures, beads were suspended in the same medium with a high density (1x106 cells mL-1) of the cells of the same species. Isolated protoplasts were also cultured in the same medium without embedding in Ca-alginate bead as controls. All protoplast cultures were incubated at 25±2°C in darkness. Protoplast cultures were subcultured every 7-10 days by replacing half of the old medium by an equal volume of fresh medium with lower osmotic pressure. Medium changes were frequently performed by gradual decreasing of concentration of the growth regulators and mannitol. Growth rate was estimated by counting the fractions of cells which divided inside of the beads.
Embryogenic callus formation and plant regeneration: After 4-5 weeks of culture, microcalli that were smaller than 1 mm in diameter appeared on the surface of the beads. When these beads were transferred to half strength MS agar medium supplemented with 2.32 μM kinetin and 2.26 μM 2,4-D, protoplast-derived embryogenic calli larger than 5 mm in diameter appeared. Embryogenic calli were transferred to a half-strength MS medium without growth regulator for maturation of somatic embryos. For germination and conversion to plantlets, somatic embryos showing bipolarity were transferred to half strength MS medium containing 14.45 μM GA3 and 4.43 μM BA and incubated under a 16/8 h (light/dark) photoperiod with a temperature of 20±2°C.
RESULTS
Induction of embryogenic callus: After 6 weeks of culture, shoot meristems on MS medium containing different growth regulators formed calli. Frequency of callus initiation and morphogenetic nature of callus varied with kind of growth regulator used (Table 1). The highest frequency of callus induction (74.3±1.5) and embryogenic callus induction (49.3±3.4) were recorded on MS medium with kinetin and 2,4-D suggests cytokinin and auxin requirement for induction of embryogenesis and growth of callus in this plant.
Selection and maintenance of embryogenic callus: During initial stages of callus development yellowish and soft calli grew from cultured explants, which after four transfers were visually distinguishable to be of two types: yellow to brownish friable and nodular embryogenic calli with shining globular structures and yellowish soft calli (Fig. 1A and B).
 | |
| Fig: 1A-M: | Plant regeneration of protoplast-derived embryogenic calli of C. pallasii subsp. haussknechtii (A) Nonembryogenic callus (B) Embryogenic callus (C) Isolated protoplasts (D) Nursed Ca-alginate beads (E) First and second cell divisions in the bead (F) Cell colony (G) Protoplast-derived embryogenic calli on solid medium (H) Globular embryo (I) Heart-shaped embryo (J) Bipolar embryo (K) Germinated embryos (L) Plantlet with corm and (M) Plants transferred to pots |
Production of embryogenic calli increased considerably with careful selection of embryogenic regions and frequent subculturing on the maintenance medium.
Isolation and culture of protoplasts: Embryogenic calli were suitable for enzyme digestion and produced the maximum yield of protoplasts, about 5-7x10-5 protoplasts g-1 of fresh weight, after 8-10 days of subculture. Isolated protoplasts from embryogenic calli were relatively uniform in size after purification (Fig. 1C), formed a cell wall within 2-3 days and were able to undergo cell divisions, which attributes to judge the quality of protoplasts.
| Table 2: | Effects of Ca-alginate and nurse culture on the growth of protoplasts of C. pallasii subsp. haussknechtii* |
 | |
*Values are the means for two experiments and are based on the percentage of cultured protoplasts. Data were taken 4-64 days after culture. Mean separation within rows by DMRT, p≤0.05. The terms have defined as cell colony>cell cluster with 8 cells; microcallus<1 mm diam and callus>5 mm diam | |
Although control protoplasts without embedding in Ca-alginate gel showed very low cell division, immobilized protoplasts (Fig. 1D) were able to divide better and form cell colonies. The combination of Ca-alginate bead and nurse culture method improved the protoplast response and reduced the budding phenomenon (Table 2). Addition of fresh medium with lower osmotic pressure during protoplast culture promoted cell division.
Embryogeniccallus formation and plantlet regeneration: After 4-5 days of subculture, first divisions of embedded protoplasts were observed and microcalli appeared after 4-5 weeks on the surface of the beads (Fig. 1E and F). Transferring of beads to an agar half strength MS medium containing 2.32 μM kinetin, 2.26 μM 2,4-D and 5.68 μM ascorbic acid increased growth rate of calli (Fig. 1G). Transferring of embryogenic calli with globular embryos to half strength MS medium without growth regulators, resulted in early maturation of embryos. All the stages of somatic embryogenesis such as globular, heart-shaped and bipolar embryos were observed (Fig. 1H-J). A few days later it was possible to isolate bipolar embryos that subsequently germinated and converted to plantlets on half strength MS medium containing 14.45 μM GA3 and 4.43 μM BA at 20±2°C under 16/8 h light/dark cycle (Fig. 1K). Some plantlets thus developed were left to grow in culture under same conditions for more weeks produced corm (Fig. 1L). Regenerated plantlets with corms were potted into peat soil after preplanting hardening process by keeping them on low nutrient level (Fig. 1M).
DISCUSSION
The results reported here showed that two types of calli (embryogenic and nonembryogenic) grew from cultured shoot meristems. Production of embryogenic callus was initially slow but careful selection of embryogenic regions and frequent subculturing on the maintenance MS medium resulted in vigorous proliferation of embryogenic callus which were utilized as source of protoplasts. Somatic embryo development in the present case was asynchronous and various stages of globular, heart-shaped and bipolar embryos could be observed simultaneously by transferring the embryogenic calli on medium lacking auxin. With the depletion of auxin, the block on the expression of those genes required for the transition to the heart stage is removed (Von Arnold et al., 2002). This protocol agrees with general mode of plant regeneration through somatic embryogensis reported in number of plants (George et al., 1992; Ahuja et al., 1994; Ebrahimzadeh et al., 2000b). The duration of subculture on the maintenance medium and the condition of embryogenic calli influenced on quality and quantity of the isolated protoplasts. The maximum yield of protoplast obtained here (5-7x10-5 protoplasts g-1 of fresh weight) was much better than those were obtained from shorter or longer duration of subculturing. This result is similar to that of C. sativus and C. cancellatus (Ebrahimzadeh et al., 2000a; Karamian and Ebrahimzadeh, 2001). The fact that immobilized protoplasts without nurse cells could divide and form a callus suggests that entrapment in Ca-alginate gel is one of the mildest immobilization procedure know, because the gelling agent itself is not toxic and the gelling process is thermo independent. Immobilization improved a protection for fragile cells such as plant protoplasts (Brodelius and Nilsson, 1980; Isa et al., 1990; Ebrahimzadeh et al., 2000a). The nurse beads showed a high growth rate and a callus was quickly formed, suggesting that the mother cells promoted the growth of protoplasts in the nurse beads. The important role of nurse culture was also reported in many species including those of Crocus earlier (Brodelius and Nilsson, 1980; Isa et al., 1990; Ebrahimzadeh et al., 2000a; Karamian and Ebrahimzadeh, 2001). In this experiment, the original medium was frequently replaced with a medium with lower osmotic pressure. Protoplast culture is usually successful when medium changes are frequently performed to allow a gradual decrease of the osmotic pressure and concentration of growth regulators. Through the use of Ca-alginate beads, this objective could be attained easily and quickly. Calli derived protoplasts on half strength MS medium without plant growth regulators showed all the stages of somatic embryo development. However, germination of somatic embryos was obtained in the presence of GA3 and BA. Plantlet regeneration through somatic embryos grew from protoplasts was achieved on the same medium at 20±2°C. Since the species of the genus Crocus grow from late autumn to early spring in nature, callus culture under low-temperature conditions was attempted (Isa et al., 1990; Ahuja et al., 1994; Ebrahimzadeh et al., 2000b; Karamian and Ebrahimzadeh, 2001). Many aspects can affect the maturation and germination of somatic embryos grew from protoplasts, such as temperature and light conditions (Tremblay and Tremblay, 1991; Firoozabady and DeBoer, 1993), age of explants (Iida et al.,1992; Brown and Watson, 1993), gelling agent, concentration of growth regulators (Morris et al., 1990; Tremblay and Tremblay, 1991) and the culture system applied (Laudenir and Sondahl, 1989; Larkin et al., 1993). The data reported here demonstrated for the first time the plant regeneration from protoplasts derived embryogenic calli of C. pallasii subsp. haussknechtii. This effective approach offers the possibility to mass multiply material that has been improved by genetic manipulation experiments.
ACKNOWLEDGMENTS
The author wish to thank Dr. M. Ranjbar for his valuable helps.
REFERENCES
- Von Arnold, S., I. Sabala, P. Bozhkov, J. Dyachok and L. Filonova, 2002. Developmental pathways of somatic embryogenesis. Plant Cell. Tissue Organ Cult., 69: 233-249.
CrossRefDirect Link - Brown, D.C.W., E.M. Watson and P.M. Pechan, 1993. Induction of desiccation tolerance in microspore-derived embryos of Brassica napus. In vitro Cell Dev. Biol. Plant, 29: 113-118.
Direct Link - Ebrahimzadeh, H., R. Karamian and M.R. Nori-Daloii, 2000. Shoot regeneration from saffron protoplasts immobilized in Ca-alginate beads. J. Sci. I. R. Iran, 11: 69-72.
Direct Link - Ebrahimzadeh, H., R. Karamian and M.R. Nori-Daloii, 2000. Somatic embryogenesis ad regeneration of plantlet in saffron, Crocus sativus L. J. Sci. I. R. Iran, 11: 169-173.
Direct Link - Firoozabady, E. and D.L. DeBoer, 1993. Plant regeneration via somatic embryogensis in many cultivars of cotton (Gossypium hirsutum L.). In vitro Cell. Dev. Biol., 29: 166-173.
Direct Link - George, P.S., S. Visvanath, G.A. Ravishankar and L.V. Venkataraman, 1992. Tissue culture of saffron (Crocus sativus L.): Somatic embryogenesis and shoot regeneration. Food Biotechnol., 6: 217-223.
Direct Link - Iida, Y., K. Watable, H. Kamada and H. Harada, 1992. Effect of abscisic acid on the induction of desiccation tolerance in carrot somatic embryos. J. Plant Physiol., 140: 356-360.
Direct Link - Isa, T., T. Ogasawara and H. Kaneko, 1990. Regeneration of saffron protoplasts immobilized in Ca-alginate beads. Jap. J. Breed., 40: 153-157.
Direct Link - Karamian, R. and H. Ebrahimzadeh, 2001. Plantlet regeneration from protoplast-derived embryogenic calli of Crocus cancellatus. Plant Cell Tissue Org. Cult., 22: 115-121.
CrossRefDirect Link - Ma, R., Y.D. Guo and S. Pulli, 2003. Somatic embryogenesis and fertile green plant regeneration from suspension cell-derived protoplasts of rye (Secale cereale L.). Plant Cell Rep., 22: 320-327.
Direct Link - Maddock, S.R., 1987. Suspension and protoplast culture of hexaploid wheat (Triticum aestivum L.). Plant Cell Rep., 6: 23-26.
Direct Link - Morris, P.C., A. Kumar, D.J. Bowles and A.C. Cuming, 1990. Osmotic stress and abscisic acid induce expression in wheat EM genes. Eur. J. Biochem., 190: 625-630.
Direct Link - Murashige, T. and F. Skoog, 1962. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant., 15: 473-497.
CrossRefDirect Link - Larkin, P.J., P.A. Davis and G.J. Tanner, 1993. Nurse culture of low numbers of Medicago and Nicotiana protoplasts using calcium alginate beads. Plant Sci., 58: 203-210.
Direct Link - Lee, B.T., K. Murdock, J. Topping, M.T.J. De-Both and Q.S. Wu et al., 1988. Isolation, culture and morphogenesis from wheat protoplasts and study of expression of DNA constructs by direct gene transfer. Plant Cell Tissue Org. Cult., 22: 233-236.
CrossRef - Tremblay, L. and F.M. Tremblay, 1991. Effect of gelling agent, ammonium nitrate and light on the development of Picea mariana (Mill) B.S.P. (black spruce) and Picea rubens Sarg. (red spruce) somatic embryos. Plant Sci., 77: 233-242.
Direct Link - Vasil, I.K., 1983. Isolation and culture of protoplasts of grasses. Int. Rev. Cytol. Suppl., 16: 79-88.
Direct Link - Vasil, I.K., 1988. Progress in the regeneration and genetic manipulation of cereal crops. Bio. Technol., 6: 397-402.
Direct Link - Vasil, I.K. and V. Vasil, 1992. Advances in cereal protoplast research. Phisiol. Plant, 85: 279-283.
Direct Link








