ABSTRACT
The reproductive activity of the mangrove prosobranch, Pachymelania fusca was studied monthly in the University of Lagos mangrove swamp, at the edge of the Lagos lagoon. The reproductive cycle was analysed from November 2003 to March 2005 by histological examination and macroscopic features of the gonads. Four gonadal stages are described: recently spawned, unripe, ripe and spawning gonads. Sex ratio obtained during this study was 1: 0.95; (51.24% males, 48.76% females). On month by month, there were three peaks when there were more females than males; October-December 2003, October-December 2004 and February-March 2005. Histological analyses revealed that reproductive activity was synchronous in both sexes, from October to March, the exact timing appearing to vary in successive years by about one month. Spawning occurred at end of rainy season and the beginning of the dry season, when the salinity of the water ranged from about 0.5-25.5 psu and surface water temperature was 29-30°C. The resting phase was April to June, during the rainy and low salinity season when the mudflat is flooded and the water turbulent. It is suggested that the species limits its breeding season to a period when there is sufficient food available and when the eggs and larvae will not be washed away.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2007.649.653
URL: https://scialert.net/abstract/?doi=pjbs.2007.649.653
INTRODUCTION
The family Melaniidae, Super family Cerithiacea, is represented in the lagoons, estuaries and mangrove swamps by Pachymelania, which is endemic to West Africa (Buchanan, 1954). The genus Pachymelania is distributed along the coast, in the estuaries and lagoons of West Africa from Senegal to Angola (Nicklés, 1950).
Pachymelania adapts perfectly to fresh water but prefers brackish water of higher salinity and is often extremely abundant in the mangrove swamps and on the mud-flats within reach of the tide, in the lagoons and river estuaries. Of the 4 species, only P. bryonensis inhabits fresh water, the other 3 species are characteristics of brackish tidal waters and mangrove swamps along the West African coast.
Pachymelania fusca (Gmelin, 1791) var. quadriseriata (Gray) is never found in the main body of the lagoon; it is restricted to the mangrove swamp and mud flat. Its distribution frequently coincides with that of the Tympanotonus fuscatus and the two are grouped together and called periwinkles in Nigeria, though they are not littorinid snails.
Most of the literature on Pachymelania is concerned with shell characteristics, geographic distribution and classification (Nicklés, 1950; Oyenekan, 1975; Plaziat, 1977). The only literature on the biology of the species is by Binder (1968) and Oyenekan (1975) who gave an account of the anatomy, ecology and systematics of Pachymelania aurita, Pachymelania fusca and Pachymelania bryonensis. Very little information is available on the reproductive cycle of P. fusca despite its abundance in the benthic community (Oyenekan, 1975) and economic importance as edible snail.
The objective of this study is to present the main features of the reproductive biology of the population of P. fusca in the University of Lagos mangrove swamp. The description is based on the observation and histological study of the male and female gonad during a 16-month period.
MATERIALS AND METHODS
Specimens of Pachymelania fusca var. quadriseriata were collected from the sampling site in the mangrove swamp of the Lagos lagoon in the University of Lagos, Nigeria ((lat N 0 6°31.049, long E 003°24.149), twice a month between November 2003 and March 2005. Individuals were collected during low tide by hand picking and collected samples were brought alive to the laboratory and maintained in lagoon water aquaria. They were measured with a caliper to the nearest 0.01 mm; specimens of size range 21-30 mm were selected, to minimize the influence of size on gonad indices. To determine the sex ratio, 50 individuals, 21-30 mm long were selected, the shell of each was broken and the colour of gonad was used to determine sex. To test the hypothesis that sex ratio was not significantly different from 1:1, a χ2-test and one-way ANOVA were carried out.
For histological analysis, fragments of the testis and ovary were fixed in aqueous Bouin, dehydrated in graded alcohol, cleared in xylene and embedded in paraffin wax. Sections were cut at 6 μm and stained in haematoxylin and eosin. Reproductive cycle stages were determined based on macro (colour and consistency) and microscopic characteristics of the gonads.
Hydrographic data (temperature and salinity) were collected from the surface water at each visit.
RESULTS
Sex ratio: A total of 1.897 individuals were collected and analysed for sex ratio during 16 months sampling months, from October 2003 to March 2005. All snails dissected had either female or male organs and the sex ratio was 1:0.95; out of these 972 (51.24%) were males and 925 (48.76%) were females. Chi squared test results indicated that the sex ratio was not significantly different from 1:1 (χ2 = 1.11; d.f. = 1; 0.29<p<0.5). On month to month basis (Fig. 1), there were three peaks when there were more females than males; October-December 2003, October-December 2004 and February-March 2005. One-way analysis of variance (ANOVA) indicated that the monthly sex ratio was not significantly different from 1:1 (p>0.05).
Gonad colour: There was considerable variation in gonad colour, from October 2003 to March 2005. The mature ovary is light green and testis is bright orange. Other colours exhibited by the ovaries are purple, dark green, brown and testes, yellow, brown-orange and brown.
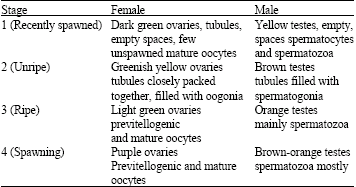
Gonadal maturation: On the basis of histological and morphological observations, four stages of gonadal development of ovary and testis are distinguishable in mature P. fusca, namely; stage 1, recently spawned; stage 2, unripe; stage 3, ripe; stage 4, spawning (Table 1).
Ovaries: Dark green ovaries (stage 1, recently spawned, Fig. 2a) were present in some of the months sampled but were predominant in the following months; February 2004 (49%), March 2004 (40%), May 2004 (82%), June 2004 (81%), July 2004 (57.5%), October 2004 (58%), February 2005 (48%) and March 2005 (60%).
Brown ovaries (stage 2, unripe, Fig. 2a) were present in some samples collected between October 2003 and March 2005 (Fig. 2a) but were predominant in October 2003 (30%), November 2003 (22.5%), January 2004 (27%) and February 2004 (28.6%).
Light green ovaries (stage 3, ripe, Fig. 2a) were many in the samples in November 2003 (31.25%), March 2004 (15%), April 2004 (16.25%), August 2004 (25%), September 2004 (39%) and December 2004 (22%) and predominant in December 2003 (50%). In the samples collected between January and March 2005, light green ovaries were found only in February (6.5%).
Females exhibiting purple ovaries (stage 4, spawning, Fig. 2a) were present in all the samples collected between October 2003 and March 2005 but were predominant from October to December 2003 (70, 46.5 and 50%). In 2004, purple ovaries were predominant in January (56%), March (45%), April (47.75%), September (61%), November (51%) and December (62%). Between January and March 2005, purple ovaries were predominant only in January (62%).
| Table 1: | Macroscopic and microscopic features of gonadal maturation stages in Pachymelania fusca in the mangrove swamp, University of Lagos |
 | |
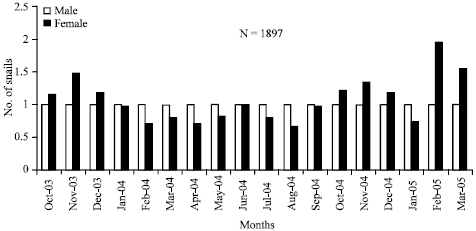 | |
| Fig. 1: | Monthly sex ratio of Pachymelania fusca |
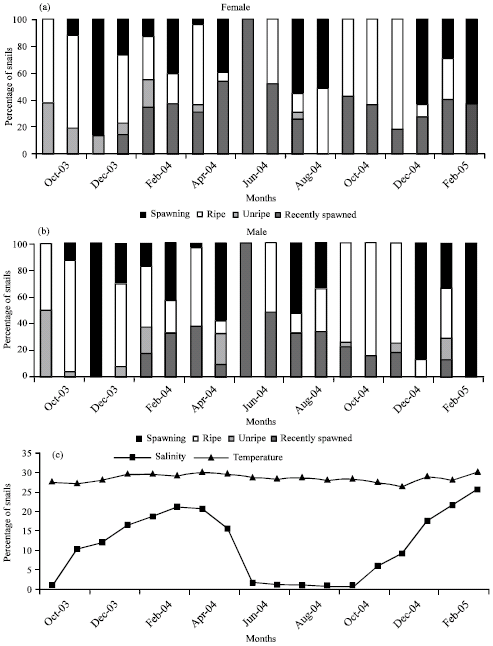 | |
| Fig. 2: | (a, b) Percentage of gametogenetic stages in Pachymelania fusca. (c) Hydographic data of surface water of the mangrove swamp, University f Lagos, Nigeria |
Testes: Yellow testis (stage 1, recently spawned, Fig. 2b) appeared in the samples in the following months; February 2004 (17%), March 2004 (32%), April 2004 (37.5%), May 2004 (9%), June 2004 (100%), July 2004 (47.5%), August 2004 (32.5%), September 2004 (30%), October 2004 (20%), November 2004 (15%), December 2004 (18%) and February 2005 (12.5%).
Brown testes (stage 1, unripe, Fig. 2b) appeared in the samples predominantly in October 2003 (50%).
Orange testes (stage 3, ripe, Fig. 2b) were present in all the months sampled but were predominant in the following months; October 2003 (50%), November 2003 (85%) January 2004 (63%), February 2004 (46%), April 2004 (60%), July 2004 (52.5%), October 2004 (78.5%), November 2004 (85%), December 2004 (75%) and February 2005 (37.5%).
Brown-orange (stage 4, spawning, Fig. 2b) testes were present in most of the months sampled but were predominant in the samples collected in the following months; December 2003 (100%), March 2004 (42%), May 2004 (58%), August 2004 (52.5%), January 2005 (86%) and March 2005 (100%).
Salinity and temperature: The surface water temperature was between 26.3 and 30°C; the lowest temperature (26.3°C) was in December 2004 and highest temperature (30°C) was in April 2004 and March 2005 (Fig. 2c). The salinity of the habitat was low between June and October (Fig. 2c) and high from October to May. The lowest salinity recorded during this study was 0.1 psu in October 2004 and the highest was 25 psu in March 2004. The mudflat dries up between December and April in most areas.
Overview of gonad maturation: Recently spawned (stage 1) females and males were present in the samples collected between March and July and females were more than the males. Salinity was between 25.5 and 1.25.
Few individuals in the samples displayed unripe gonad (stage 2); unripe females were more in the samples of October to February and unripe males in October, February and May respectively
There were not too many females displaying ripe ovary (stage 3) in the samples; they were predominant in samples between November and April (salinity, 6-20.5) and August to September (salinity, 1-0.5). Males with ripe testes were predominant in the samples between August and March (salinity, 1-25.5).
Spawning (stage 4) individuals were available in the samples all year round; females were predominant in September to February (salinity, 0.5-21.5) and males October to March (salinity 0.5-25.5).
DISCUSSION
Although Pachymelania fusca is dioecious, size distribution does not indicate that there is external sexual dimorphism, therefore, sex is only determined by the appearance and colour of the gonad. The population sex ratio for P. fusca was almost 1:1 during the period of observation. There were however three peaks when there were more females than males. Although the difference was not significant, the 3 peaks correspond to the period when P. fusca were mature and spawning. The presence of juvenile in the tanks in July is a confirmation that species spawned before July, therefore recruitment probably occurs in the natural habitat from July.
Monthly changes in the gonad showed that spermatogenesis is synchronous with oogenesis and sexually matured individuals were obtained through out the year, however, the number of individuals with ripe ovaries and testis were more between August and February 2004 and August to March 2005, (a difference of one month), this indicates that the species probably has a defined annual cycle of gametogenesis and a single spawning period of September to February for females and October to March for males.
The result of the salinity confirms observations by Hill and Webb (1958), Sandison and Hill (1966) and Olaniyan (1969) that there are two salinity seasons per annum in the Lagos lagoon system. The salinity is low in the months of May to October (rainy season) due to influx of freshwater from the rivers, storm water run-offs and rainwater and high from December to April (dry season). The result shows that P. fusca occurs naturally in waters with very low salinity between June and November, which have a large seasonal fluctuation. Egonmwan (1985) reported that the sympatric species Tympanotonus fuscatus var. radula is euryhaline, but best able to osmoregulate in lower salinities up to 17 psu.
The temperature was high all year round and typical for a tropical climate so may not affect maturation of the gonad to any appreciable extent.
The annual pattern of reproductive cycle of P. fusca may be controlled by the interaction between endogenous factors and environment. Temperature, light salinity, tidal period and food have been invoked as exogenous factors in temperate coastal waters for many bivalves (Harvey and Gage, 1995). Spawning occurred in dry season (December to March) (salinity, 9.2-25.5 psu). From May to July (15.5-1.25 psu), the individuals were in resting phase. Seasonal changes in both male and female snails was reported in the brackish water Rissooidea, Iravadia sakaguchii (Kobayashi and Wada, 2004). Temperature may not a limiting factor in the tropical coastal region as it is uniform through out the year; rather, the availability of suitable food supply during the months prior to spawning may be a more potent determinant of spawning time (Harvey and Gage, 1995). The mud flat in the University of Lagos where this study was conducted dries up in most areas during the height of dry season (February-April) and available food may diminish. Also turbulent waters with fast bottom currents have sandy bottoms containing little organic content.
The present investigation seems to indicate that the main factors controlling reproduction in P. fusca are salinity, water and current. P. fusca, a euryhaline species can live in fresh water but prefers brackish water of rather high salinity water of the lagoon. During the breeding season, salinity increases from 0.50 psu in October to 25.5 psu in March. According to Egonmwan (1986), salinity in the mangrove swamps around the University of Lagos is high between February and April and low from June to November. The habitat dries up between February and April, during the dry season. The rains begin in late April and downpour is heaviest in June, July and August. During the rainy season, the mudflat is flooded with rainwater, the salinity of the water in mud flat is reduced and there is no breeding during this period probably to prevent the fertilized eggs and developing embryo being washed away by the flood current. Salinity is also a major factor controlling the distribution of the sympatric species, Tympanotonus fuscatus var. radula, a prosobranch, in the same habitat (Binder, 1968; Pauly, 1975; Egonmwan, 1985).
The annual cycle of reproduction in P. fusca coincides with the end of rainy season and the beginning of the dry season in Lagos and maturing of gonad and spawning is associated with increasing salinity, a period when the environmental conditions are suitable for the pelagic larvae. Spawning at the end of the raining season and beginning of dry season would therefore appear to have an adaptive value to a benthic species like P. fusca.
REFERENCES
- Harvey, R. and J.D. Gage, 1995. Reproduction and recruitment of Nuculoma tenuis (Bivalvia: Nuculoida) from Loch Etive, Scotland. J. Moll. Stud., 61: 409-419.
Direct Link - Hill, M.B. and J.E. Webb, 1958. The ecology of the Lagos lagoon. Part 11: The topography and physical features of the Lagos harbour and Lagos lagoon. Phil. Trans. R. Soc. Lond., B, 241: 319-333.
Direct Link - Kobayashi, Y. and K. Wada, 2004. Growth, reproduction and recruitment of the endangered brackish water snail Iravadia (Fairbankia) sakagushii (Gastropoda: Iravadiidae). Molluscan Res., 24: 33-42.
Direct Link








