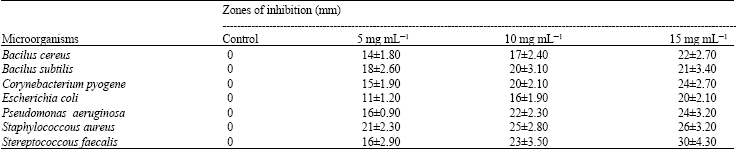
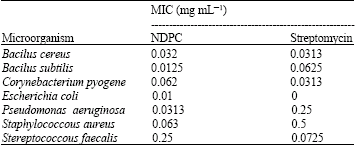
Research Article
Antibacterial Effect of N-Naphtylen Diamine Platinum (II) Choloride as Novel Compound
Department of Biology, Faculty of Science, Cell and Molecular Laboratory, Bu-Ali Sina University, Iran
Seyed Javad Sabounchi
Department of Chemistry, Faculty of Science, Bu-Ali Sina University, Hamedan, Iran
Vida Jodaian
Department of Chemistry, Faculty of Science, Bu-Ali Sina University, Hamedan, Iran