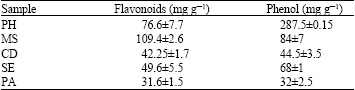Research Article
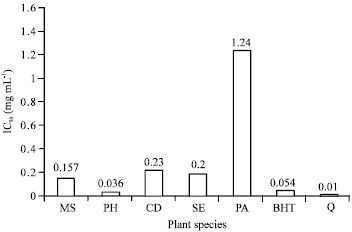
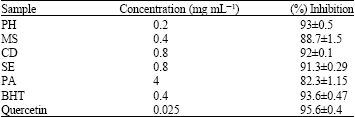
In vitro Antioxidant Activity of Polygonium hyrcanicum, Centaurea depressa, Sambucus ebulus, Mentha spicata and Phytolacca americana
Department of Medicinal Chemistry, Faculty of Pharmacy, Mazandaran University of Medical Sciences, Sari, Iran
Fereshteh Pourmorad
Department of Medicinal Chemistry, Faculty of Pharmacy, Mazandaran University of Medical Sciences, Sari, Iran
Naghi Shahabimajd
Department of Physiology and Pharmacology, Faculty of Medicine, Mazandaran University of Medical Sciences, Sari, Iran
Kami Shahrbandy
Department of Medicinal Chemistry, Faculty of Pharmacy, Mazandaran University of Medical Sciences, Sari, Iran
Rasa Hosseinzadeh
Department of Medicinal Chemistry, Faculty of Pharmacy, Mazandaran University of Medical Sciences, Sari, Iran