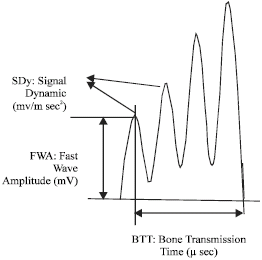
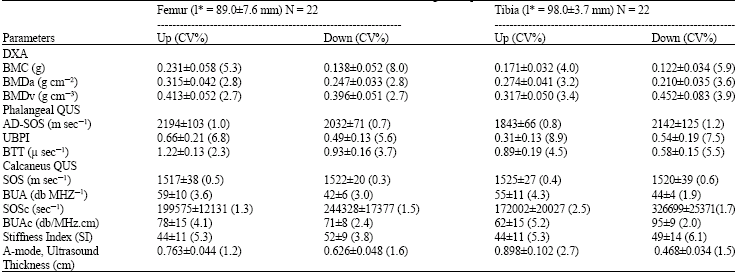
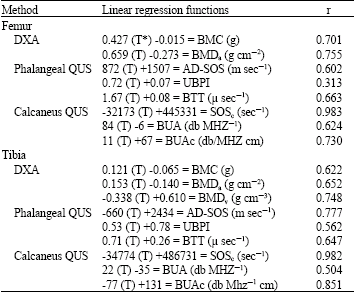
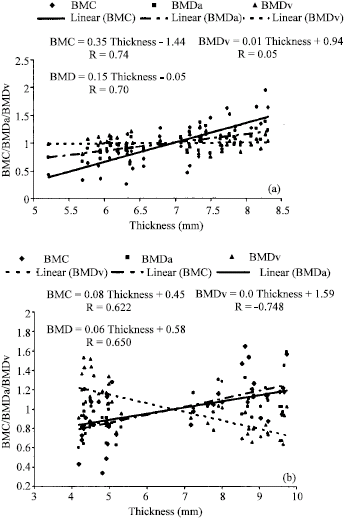
Research Article
Influence of Bone Thickness on Densitometric and Ultrasonic Parameters, an in vivo Study
Department of Medical Physics, Tarbiat Modarres University,Tehran, Iran
M. R. Dadras
Department of Medical Physics, Tarbiat Modarres University,Tehran, Iran
B. Larijani
Metabolism and Endocrine Research Center, Tehran Medical Sciences University, Tehran, Iran