ABSTRACT
Extracts of 42 medicinal plants used for the treatment of anaemia, diabetes, AIDS, malaria and obesity were screened for phytochemical substances and antioxidant potentials. The plant extracts were prepared as hydrolysed (for total antioxidant) and non-hydrolysed (for free antioxidant). Extracts were analysed using three different assay methods for antioxidant analysis: Folin (Folin Ciocalteu reagent), FRAP (Ferric Reducing Antioxidant Power) and DPPH (1,1-diphenyl-2-picrylhydrazyl). The leaves of Alchornea cordifolia showed the highest antioxidant properties as determined by both Folin and FRAP free antioxidant, followed by Dacryodes edulis and Ocimum basilicum in FRAP and by Dacryodes edulis, Harungana madagascariensis for Folin and DPPH method. For total antioxidant activity, Alchornea cordifolia was ranked first followed by Dacryodes edulis, Harungana madagascariensis, Ocimum basilicum for the FRAP method while for the Folin method Harungana madagascariensis occupied the first position followed by Cylicodiscus gabunensis, Ocimum basilicum, Coleus coprosifolius, Alchornea cordifolia, Dacryodes edulis. All the plants show some antioxidant activity irrespective of the method used. The phytochemical studies revealed the presents of important bioactive constituents with antioxidant activity that may have some medicinal properties.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2007.537.544
URL: https://scialert.net/abstract/?doi=pjbs.2007.537.544
INTRODUCTION
Traditional healers and their plant medicines provide the only health care to the majority of people in a curative rather than a preventive approach in the developing countries for common ailments. The ready availability and economy of plants as direct therapeutic agents make plants more attractive when compared to modern medicine (Agbor and Ngogang, 2005; Agbor et al., 2005a). As a result, vast literature now exist on the use of traditional medicine with botanist reporting description of plants used for different disease treatments, the phytochemist on the chemical constituents and the pharmacologist on the effectiveness of particular plant compound or extracts. In the Cameroonian pharmacopoeia, several plants of folkloric medicine are used in the treatment of diseases such as malaria, diabetes, obesity, anaemia and opportunist infections of the AIDS.
The knowledge of traditional medicinal plant preparations is orally transmitted from one generation to another, a procedure that involves the risk of loss of essential information. To ensure the continuity of this precious cultural heritage, an increasing number of ethnobotanic inventories have been established (Iwu, 1993; Adjanohoun et al., 1996; Atindehou et al., 2004). However, there is still reluctance to adopt traditional medicine in Cameroon because they lack scientific evidence, particularly about their cocktail nature, safety and usefulness in more than one type of disease conditions.
Many natural products have been reported to contain large amounts of antioxidants other than vitamin C, E and carotinoids (Javanmardi et al., 2003). These antioxidants play a role in delaying, intercepting, or preventing oxidative reactions (Vilioglu et al., 1998) catalysed by free radicals. This antioxidant activity may be mainly due to the presents of phenolic components such as flavonoids (Pietta, 1998), phenolic acids and phenolic diterpenes (Shahidi et al., 1992). Free radicals, Reactive Oxygen Species (ROS) and Reactive Nitrogen Species (RNS) are associated with many pathological conditions such as inflammation, metabolic disorders, cells ageing, atherosclerosis and carcinogenesis (Roback et al., 1988; Ames et al., 1993). According to Yildrim et al. (2001), the ROS are involved in more than one hundred diseases including, malaria, acquired immunodeficiency syndrome, diabetes, anaemia and cardiovascular diseases.
Thus, it is necessary to find out if medicinal plants could provide the antioxidant substances that may help to modulate oxidative stress related disorders. In order to do this, the present study was designed to evaluate 42 medicinal plants commonly used in Cameroon for phytochemical composition and antioxidant capacity.
MATERIALS AND METHODS
Plants material: Some of the medicinal plants were harvested from their natural habitat while others were bought from the local markets in Dschang and Douala, Cameroon and identification was confirmed in the National herbarium Yaoundé. After air-drying, the samples were blended into powder. Both hydrolysed (total antioxidant) and non-hydrolysed (free antioxidant) extracts were prepared as earlier described by Agbor et al. (2005b).
Phytochemical screening: The methods described by Trease and Evans (1983) were used for phytochemical screening of the medicinal plants extracts for the presents of bioactive compound. This is a qualitative test based on the colour change as an indication of a positive test.
Phenol content: The phenolic content of both extracts were measured at 750 nm using Folin-Ciocalteu (Sigma Chemical Co St Louis, MO) reagent diluted 10 times before use with catechin as standard. Optical density was read after 20 min of incubation.
DPPH scavenging activity: Scavenging activity against the DPPH (1,1-Diphenyl-2-Picrilhydrazyl) free radical was studied as follows: 20 μL of extract was introduced into 2 mL of a methanolic solution of DPPH (0.3 mM) and kept in the dark for 30 min. The extract was replaced by methanol for the control and catechin for the standard. The absorbance was then spectrophotometrically read at 517 nm and the antioxidant content and extract percentage inhibition of DPPH radical were calculated as earlier described (Yen and Duh, 1994).
Ferric reducing antioxidant power: The Ferric Reducing Antioxidant Power (FRAP) of extracts was determined using the method of Benzie and Strain (1996). The FRAP reagent consisted of ten part acetate buffer (300 mM pH 3,6), one part of TPTZ (10 mM in 400 mM of HCl, Sigma) and one part of ferric chloride (10 mM).
Statistical analysis: Measurements of absorbance were made in triplicate and the results presented as mean±standard deviation. Student t-test was employed to evaluate the difference between free and total phenols of the medicinal plant extract (p<0.001). The relation between the methods was established by applying Pearson product moment correlation (p<0.05). The software SigmaStat (Systat software Richmond, CA) version 3.01 was used for this analysis.
RESULTS
The different medicinal plants harvested and their medicinal values according to traditional healers are presented in Table 1. Of the 42 medicinal plants selected, seven were anti-anaemic, seven anti-diabetic, thirteen anti-malarial, eight anti-HIV/AIDS and seven anti-obese as used by the traditional healers. Table 2 presents the results of the phytochemical screening that revealed the presence of important antioxidant bioactive molecules such as tannins, flavonoïdes and phenols.
The results of the antioxidant capacity of each sample as analysed by the various methods are presented in Table 3. The free and total antioxidant capacity were measured using both the Folin Ciocalteu (Folin) and FRAP reagents while DPPH was used to determine free antioxidant capacity and radical scavenging activity. A significant difference (p<0.001) was obtained between free and total antioxidants with total antioxidants being comparatively higher than free antioxidants in most of the plants studied. The exception of this case were A. indica, P. guajava, L. macrophyllum, E. speciosa, P. americana, B. pilosa, A. gangetica, Crinum sp., D. déisteliana where no significant difference (p>0.05) was observed between free and total antioxidant capacity.
The analysis of the scavenging activity by DPPH method is presented in Table 3 with A. cordifolia, D. edulis, H. madagascariensis giving the greatest scavenging activity (more than 90% inhibition) while C. ciliata and E. indica had less activity.
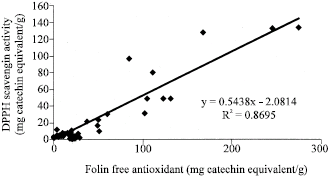
Figure 1 to 6 summarises the relationship between the Folin, FRAP and DPPH antioxidant activity. A significant correlation (p<0.05) was observed between free and total Folin antioxidant, free and total FRAP antioxidant on one hand; between Folin and FRAP, FRAP and DPPH and Folin and DPPH on the other hand.
| Table 1: | Medicinal plants harvested |
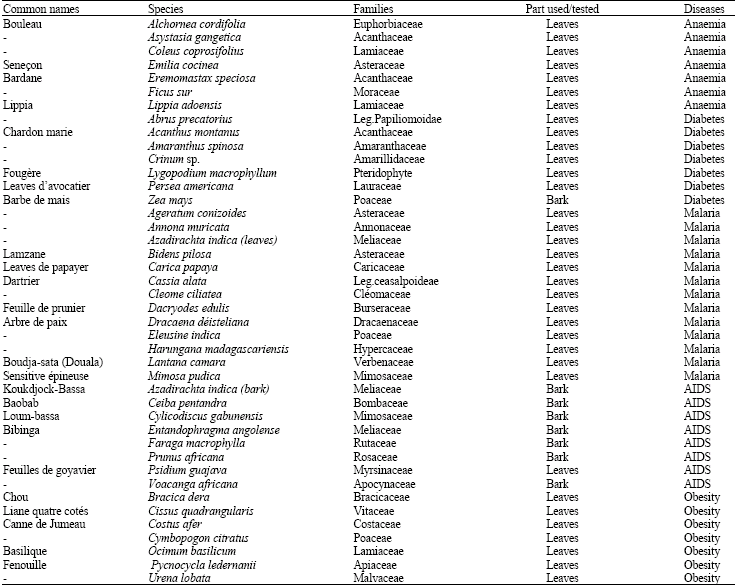 | |
| Table 2: | Phytochemical screening of some Cameroonian plants |
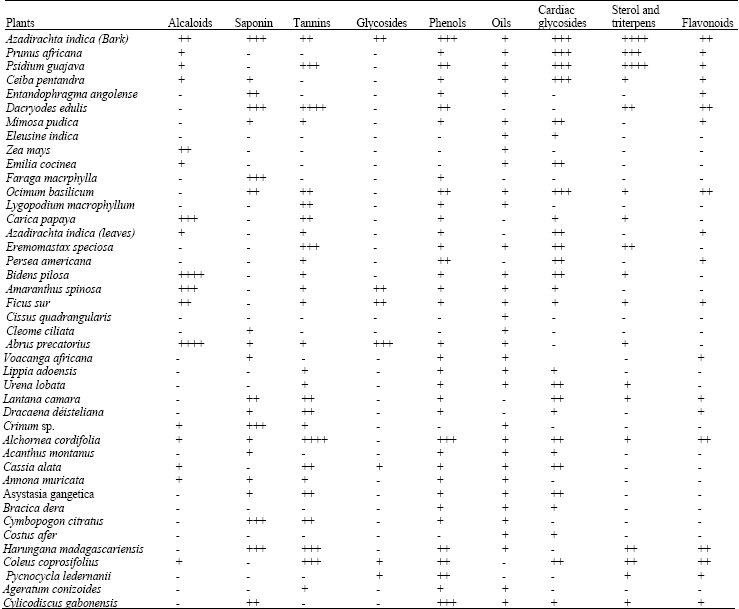 | |
- = Negative reaction, + = Positive reaction | |
| Table 3: | Free and total antioxidant capacity (mg of catechin equivalent/g of dry weight) by FRAP, Folin and DPPH methods |
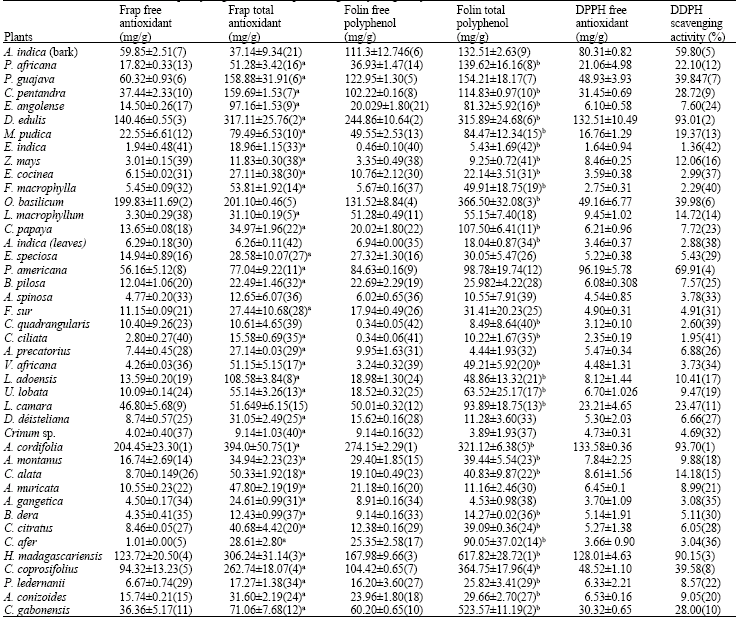 | |
aSignificantly different from FRAP free phenol, bSignificantly different from free, ( ) ranking | |
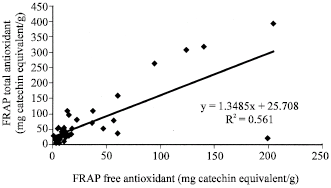 | |
| Fig. 1: | Correlation analysis, between FRAP free and total antioxidant capacities of the studied samples |
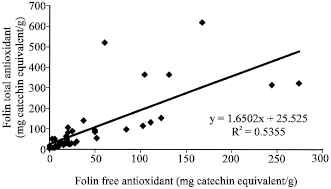 | |
| Fig. 2: | Correlation analysis between Folin free and total antioxidant capacities of the studied samples |
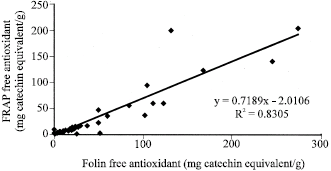 | |
| Fig. 3: | Correlation analysis, between Folin free and FRAP free antioxidant capacities of the studied samples |
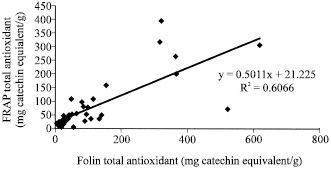 | |
| Fig. 4: | Correlation analysis, between Folin total and FRAP total antioxidant capacities of the studied samples |
 | |
| Fig. 5: | Correlation analysis between Folin free antioxidant capacity and DPPH scavenging activity of the studied samples between |
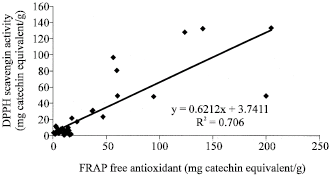 | |
| Fig. 6: | Correlation analysis, between FRAP free antioxidant capacity and DPPH scavenging activity of the studied samples |
DISCUSSION
Over the years, the research on medicinal plants to elucidate the mechanism of action and to justify their claims by traditional healers has been on the increase. An angle of this research has been the study of the bioactive components and antioxidant properties of these plants an area in which very little has been done in Cameroon. Herbs, spices, vegetables, fruits, medicinal plants, teas have been reported to have biological effects such as antioxidant properties in management and treatment of oxidative stress related disorder (Agbor et al., 2005b; Vinson et al., 1995a, b, 2001). Increased consumption of fruits and vegetables is associated with a lower risk of degenerative diseases that come with aging such as cancer, cardiovascular disease, cataracts and brain and immune dysfunction (Ames et al., 1993). These positive influences have been attributed to natural antioxidant phytochemicals. It has been shown that plants phenols such as flavonols, anthocyanins and phenylpropanoids might act as antioxidants or as agents of other mechanisms contributing to cardioprotective action (Vinson et al., 1995a, b; Wang et al., 1997; Gorinstein et al., 2004).
Folin, measures the ability of the extracts (phenolic content) to reduce the chromogen compound phosphomolybdic -tungstic of the Folin-Ciocalteu reagent. The principal antioxidant constituents of natural products are phenolic compounds that are comprised of phenolic acids and flavonoids (Kähkönen et al., 1999). Phenolic antioxidants are potent free radical terminators (Shahidi et al., 1992). They donate hydrogen to free radicals and hence, break the reaction of lipid oxidation at the initiation step (Gülçin et al., 2004). Phenolic acids such as caffeic acid, ferulic acid and vanillic acid have been implicated as natural antioxidants in fruits vegetables and other plants (Javanmardi et al., 2003; Larson, 1988). The high potential of phenolics to scavenge free radicals may be due to the many phenolic hydroxyl groups (Sawa et al., 1999) they posses.
DPPH forms a stable molecule on accepting an electron or a hydrogen atom and thus has application in the determination of radical scavenging activity of natural products (Jun et al., 2004). In situ, free radicals like polyaromatic hydrocarbon cations have been partly linked with carcinogenesis (Yen and Chen, 1995). Free radicals have been reported to be responsible for the destructive effects on protein in cataract formation, oxidative damage to DNA in the formation of certain cancers and lipid oxidative damage in the occurrence and progression of vascular diseases (Langsethm, 1995; Alho and Leinonen, 1999). Aerobic respiration, stimulated polymorphonuclear leukocytes, macrophages and peroxisomes are the main endogenous sources of most of the oxidants produced by cells (Alho and Leinonen, 1999; Niki, 2001). Thus, plants with free radical scavenging activity will prevent damage to DNA, lipid peroxidation, hence the progression of vascular diseases. In the present research, many of the plants appeared to have high DPPH scavenging activity, classified as follows: A. cordifolia, D. edulis, H. madagascariensis, P. americana, A. indica, O. basilicum, P. guajava and C. coprosifolius.
FRAP measures the ferric reducing ability of the antioxidant molecule at a low pH, forming an intense blue colour as the ferric tripyridyltriazine (Fe3+-TPTZ) complex is reduced to the ferrous (Fe2+) form and absorbance measured at 593 nm (Singleton et al., 1999).
Though a significant correlation (p<0.05) was observed between the different methods used in this analysis, the ranking was not the same making it difficult to know which method is best fit for antioxidant analysis. However, since the Folin Ciocalteu reagent measures the phenolic concentration, DPPH measured the radical scavenging activity and FRAP measures the ferric reducing antioxidant power, these differences are bound to occur.
In the classification of FRAP free antioxidant, A. cordifolia is the best antioxidant source followed by O. basilicum, D. edulis, H. madagascariensis, C. afer came in the last position, preceeded by Eleusine indica. On the other hand, in the classification of Folin free phenol antioxidant, A. cordifolia was still first followed by D. edulis, H. madagascariensis, O. basilicum. C. quadrangularis tailed the classification, preceded by C. ciliata. In FRAP total antioxidant classification, A. cordifolia was still classified first, followed by D. edulis, H. madagascariensis, C. coprosifolius, A. indica ended the classification preceded by L. macrophyllum. We also observed that H. madagascariensis occupied the first position in Folin total phenol antioxidant followed by C. gabonensis, O. basilicum, C. coprosifolius, A. cordifolia, D. edulis. The high antioxidant capacity of A. cordifolia may be responsible for its antidiarrhoeal activity earlier reported (Agbor et al., 2004). The antioxidant capacity of plants and foods reported in literature are generally determined by the hydro-organic extracts, which does not take into account the large quantity of compounds that remain associated with food fibres, sugars and others indigestible compounds (Pérez-Jiménez and Saura-Calixto, 2005). However, the non-extractable antioxidants can be released in the human intestine, which means that the results of the antioxidant capacity obtained just with the aqueous or organic extract would be limited, because the quantity of antioxidants released in the gastro-intestinal tract could differ qualitatively and quantitatively from that awaited from measurements of the usual aqueous or organic extracts. This supports the importance to study the total antioxidant capacity through acid extraction (hydrolysis) in order to obtain in addition to the extractible antioxidant capacity of the aqueous or organic extracts, the non-extractible antioxidant capacity of the hydrolysed extract.
The present study has demonstrated that medicinal plants could be a good source of antioxidant substances as determined by three methods. All the medicinal plants studied show some antioxidant activity irrespective of the method used for the analysis. It was also shown that the hydrolysed extracts (which liberate bound polyphenols, i.e., total antioxidant) had significantly higher antioxidant activity than the non-hydrolysed extracts (unbound polyphenols only, i.e., free antioxidant). This finding is in accordance with earlier studies (Agbor et al., 2005b; Vinson et al., 1995a, b). The medicinal plants studied are used in traditional medicine for the treatment of malaria, anaemia, obesity, diabetes and opportunistic infections of HIV/AIDS that are related to oxidative stress. Their antioxidant activity may be a contributing factor to their therapeutic applications.
REFERENCES
- Agbor, G.A., T. Leopold and N.Y. Jeanne, 2004. The antidiarrhoeal activity of Alchornea cordifolia leaf extract. Phytother. Res., 18: 873-876.
CrossRefDirect Link - Agbor, A.G., E.O. Oben and J.Y. Ngogang, 2005. Haematinic activity of Hibiscus cannabinus. Afr. J. Biotechnol., 4: 833-837.
Direct Link - Agbor, A.G., E.O. Oben, Y.J. Ngogang, C. Xinxing and J.A. Vinson, 2005. Antioxidant capacity of some herbs/spices from Cameroon: A comparative study of two methods. J. Agric. Food Chem., 53: 6819-6824.
Direct Link - Alho, H. and J. Leinonen, 1999. Total antioxidant activity measured by chemiluminescence method. Methods Enzymol., 299: 3-15.
Direct Link - Ames, B.N., M.K. Shigenaga and T.M. Hagen, 1993. Oxidants, antioxidants and the degenerative diseases of aging. Proc. Nat. Acad. Sci. USA, 90: 7915-7922.
CrossRefDirect Link - Atindehou, K.K., C. Schmid, R. Brun, M.W. Kone and D. Traore, 2004. Antitrypanosomal and antiplasmodial activity of medicinal plants from Cote d'Ivoire. J. Ethnopharmacol., 90: 221-227.
CrossRefDirect Link - Benzie, I.F.F. and J.J. Strain, 1996. The ferric reducing ability of plasma (FRAP) as a measure of "antioxidant power": The FRAP assay. Anal. Biochem., 239: 70-76.
CrossRefPubMedDirect Link - Gorinstein, S., Z. Zachwieja, E. Katrich, E. Pawelzik, R.R., Haruekit, S. Trakhtenaerg and O.M. Belloso, 2004. Comparison of the contents of the main antioxidant compounds and the antioxidant activity of white grapefruit and his new hybrid. Lebensm-Wiss U-Technol., 37: 337-343.
Direct Link - Gulcin, I., S. Beydemir, H.A. Alici, M. Elmastas and M.E. Buyukokuroglu, 2004. In vitro antioxidant properties of morphine. Pharmacol. Res., 49: 59-66.
CrossRefPubMedDirect Link - Javanmardi, J., C. Stushnoff, E. Locke and J.M. Vivanco, 2003. Antioxidant activity and total phenolic content of Iranian Ocimum accessions. Food Chem., 83: 547-550.
CrossRefDirect Link - Jun, M., U. Tohru, L. Jianzhang and F. Takeshi, 2004. Identification and evaluation of antioxidant activities of Bamboo extracts. For. Stud. China, 6: 1-5.
Direct Link - Kahkonen, M.P., A.I. Hopia, H.J. Vuorela, J.P. Rauha, K. Pihlaja, T.S. Kujala and M. Heinonen, 1999. Antioxidant activity of plant extracts containing phenolic compounds. J. Agric. Food Chem., 47: 3954-3962.
CrossRefPubMedDirect Link - Larson, R.A., 1988. The antioxidants of higher plants. Phytochemistry, 27: 969-978.
CrossRefDirect Link - Sawa, T., M. Nakao, T. Akaike, K. Ono and H. Maeda, 1999. Alkylperoxyl radical scavenging activity of various flavonoids and other phenolic compounds: Implications for the antitumor promoter effect of vegetables. J. Agric. Food Chem., 47: 397-402.
Direct Link - Shahidi, F., P.K. Janitha and P.D. Wanasundara, 1992. Phenolic antioxidants. Crit. Rev. Food Sci. Nutr., 32: 67-103.
CrossRefDirect Link - Singleton, V.L., R. Orthofer and R.M. Lamuela-Raventos, 1999. Analysis of Total Phenols and Other Oxidation Substrates and Antioxidants by Means of Folin-Ciocalteu Reagent. In: Methods in Enzymology, Burslem, G.L. (Ed.), Academic Press, Cambridge, Massachusetts, ISBN: 9780121822002, pp: 152-178.
CrossRefDirect Link - Trease, G.E., 1983. Pharmacognosy. 12th Edn., Baillière Tindall, United Kingdom, ISBN: 9780721609386, Pages: 812.
Direct Link - Velioglu, Y.S., G. Mazza, L. Gao and B.D. Oomah, 1998. Antioxidant activity and total phenolics in selected fruits, vegetables and grain products. J. Agric. Food Chem., 46: 4113-4117.
CrossRefDirect Link - Vinson, J.A., J. Jang, Y.A. Dabbagh, M.M. Serry and S. Cai, 1995. Plant phenols exhibit lipoprotein-bound antioxidant activity using an in vitro model for heart disease. J. Agric. Food Chem., 43: 2798-2799.
Direct Link - Vinson, J.A., Y.A. Dabbagh, M.M. Serry and J. Jang, 1995. Plant flavonoids, especially tea flavonols, are powerful antioxidants using an in vitro oxidation model for heart disease. J. Agric. Food Chem., 43: 2800-2802.
Direct Link - Vinson, J.A., J. Proch and P. Bose, 2001. Determination of the quantity and quality of polyphenol antioxidant in food and beverages. Methods Enzymol., 335: 103-114.
Direct Link - Wang, H., G. Cao and R.L. Prior, 1997. Oxygen radical absorbing capacity of anthocyanins. J. Agric. Food Chem., 45: 304-309.
CrossRefDirect Link - Yen, G.C. and P.D. Duh, 1994. Scavenging effect of methanolic extracts of peanut hulls on free-radical and active-oxygen species. J. Agric. Food Chem., 42: 629-632.
CrossRefDirect Link - Yen, G.C. and H.Y. Chen, 1995. Antioxidant activity of various tea extracts in relation to their antimutagenicity. J. Agric. Food Chem., 43: 27-32.
CrossRefDirect Link - Yildrim, A., M. Oktay and V. Bilalolu, 2001. The antioxydant activity of the leaves of Cydonia vulgaris. Turk. J. Med. Sci., 31: 23-27.
Direct Link








