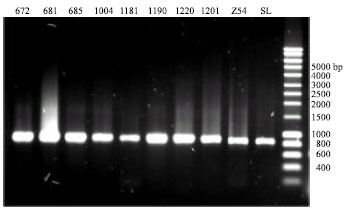
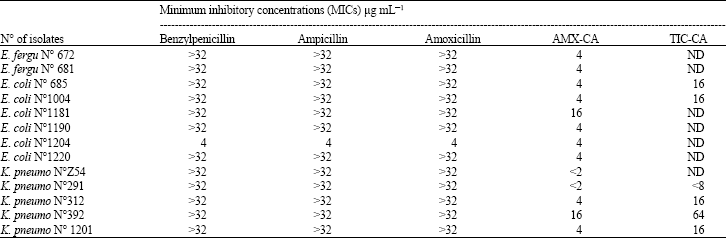
Research Article
Major Enzymatic Factors Involved in Bacterial Penicillin Resistance in Burkina Faso
Laboratoire de Biochimie et Chimie Appliquées (LABIOCA), Unité de Formation et de Recherche en Sciences de la Vie et de la Terre, Université de Ouagadougou 03 BP 7021 03 Ouagadougou, 03, Burkina Faso
Martin Kiendrebeogo
Laboratoire de Biochimie et Chimie Appliquées (LABIOCA), Unité de Formation et de Recherche en Sciences de la Vie et de la Terre, Université de Ouagadougou 03 BP 7021 03 Ouagadougou, 03, Burkina Faso
Aline Lamien
Laboratoire de Biochimie et Chimie Appliquées (LABIOCA), Unité de Formation et de Recherche en Sciences de la Vie et de la Terre, Université de Ouagadougou 03 BP 7021 03 Ouagadougou, 03, Burkina Faso
Jean-Denis Docquier
CIP/Enzymology (Université de Liège) Institute of Chemistry B6 Sart-Tilman B4000 Liège, Belgium
Jacques Simpore
Laboratoire de Biochimie et Chimie Appliquées (LABIOCA), Unité de Formation et de Recherche en Sciences de la Vie et de la Terre, Université de Ouagadougou 03 BP 7021 03 Ouagadougou, 03, Burkina Faso
Odile Germaine Nacoulma
Laboratoire de Biochimie et Chimie Appliquées (LABIOCA), Unité de Formation et de Recherche en Sciences de la Vie et de la Terre, Université de Ouagadougou 03 BP 7021 03 Ouagadougou, 03, Burkina Faso