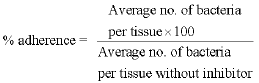Research Article
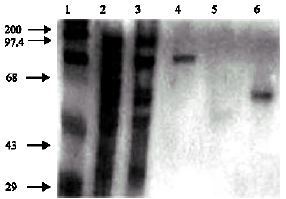
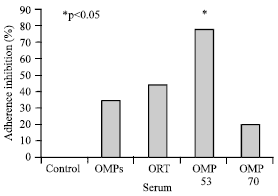
The Role of Outer Membrane Proteins of Ornithobacterium rhinotracheale in Attachment to Chicken Tracheal Epithelium
Department of Pathobiology, School of Veterinary Medicine, Shahid Chamran University, Ahwaz, Iran
M.R. Seyfi Abad Shapouri
Department of Pathobiology, School of Veterinary Medicine, Shahid Chamran University, Ahwaz, Iran
M. Jamshidian
Department of Pathobiology, School of Veterinary Medicine, Shahid Chamran University, Ahwaz, Iran
M. Mayahi
Department of Clinical Sciences, School of Veterinary Medicine, Shahid Chamran University, Ahwaz, Iran
M. Ghaforian
Department of Immunology, Medical College, Jundi Shapour Medical Sciences University, Ahwaz, Iran