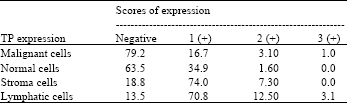Research Article
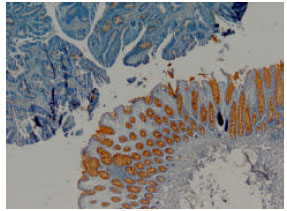
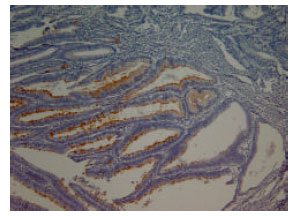
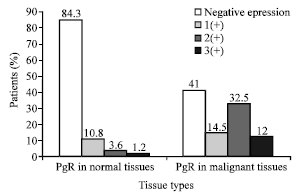
Progesterone Receptor Positive Colorectal Tumors Have Lower Thymidine Phosphorylase Expression: An Immunohistochemical Study
Department of Pathology, School of Medicine, Tehran University of Medical Sciences, Tehran, Iran
Sepideh Arbabi Bidgoli
Department of Toxicology and Pharmacology, Pharmaceutical Sciences Unit, Islamic Azad University, Iran
Mehri Mohammadi Ziyarani
Department of Toxicology and Pharmacology, Pharmaceutical Sciences Unit, Islamic Azad University, Iran
Marjan Shariatpanahi
Department of Toxicology and Pharmacology, Pharmaceutical Sciences Unit, Islamic Azad University, Iran
Farid Azmoodeh Ardalan
Department of Pathology, School of Medicine, Tehran University of Medical Sciences, Tehran, Iran