Research Article
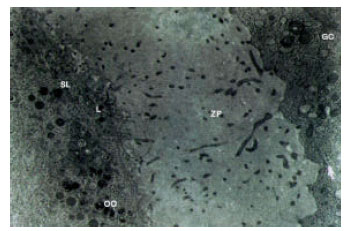
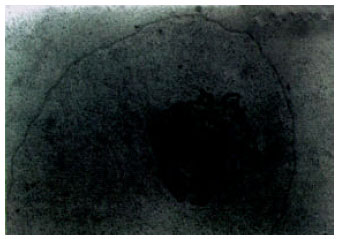
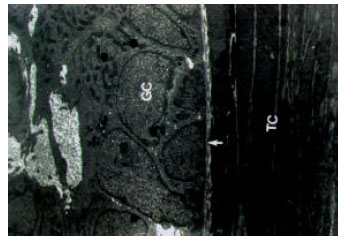
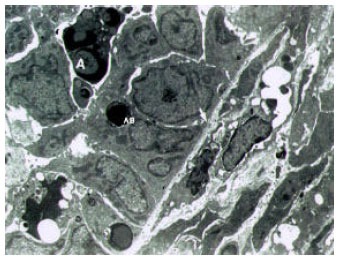
Ultrastructural Alterations and Occurrence of Apoptosis in Developing Follicles Exposed to Low Frequency Electromagnetic Field in Rat Ovary
Center of Drug Applied Research, Tabriz University of Medical Sciences, Iran
Jafar Soleimani Rad
Department of Histology and Embryology, Tabriz University of Medical Sciences, Iran