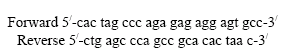Research Article
One Common Polymorphism of Cholesteryl Ester Transfer Protein Gene in Iranian Subjects With and Without Primary Hypertriglyceridemia
Department of Clinical Biochemistry, School of Medicine, Hamedan University of Medical Sciences, Hamedan, Iran
Mohsen Firoozrai
Department of Clinical Biochemistry, School of Medicine, Iran University of Medical Sciences, Tehran, Iran
Abdolvahhab Ehsani Zonouz
Department of Clinical Biochemistry, School of Medicine, Iran University of Medical Sciences, Tehran, Iran
Hassan Radmehr
Department of Cardiac Surgery, Emam Khomeini Medical Center, Tehran University of Medical Sciences, Tehran, Iran
Abbas Zavarehee
Tehran Shahid Rajaii Cardiovascular Medical Center, Tehran, Iran
Max Paoli
School of Pharmacy and Center for Biomolecular Science, University of Nottingham, Nottingham, UK