Research Article
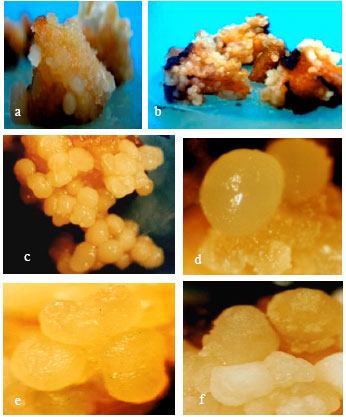
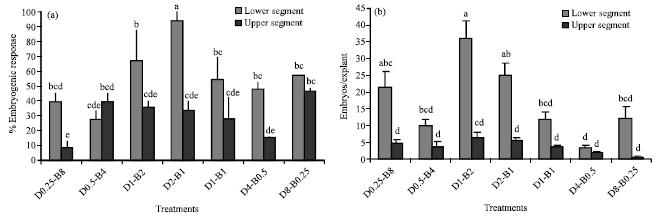
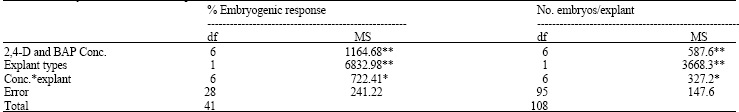
Effects of Different Concentrations of 2,4-D and BAP on Somatic Embryogenesis Induction in Saffron (Crocus sativus L.)
Department of Biology, Science Faculty, Alzahra University, Tehran, Iran
A. V. Azghandi
Department of Tissue Culture and Gene Transformation, Agricultural Biotechnology Research Institute of Iran, P.O. Box 31535-1897, Karaj, Iran
A. Saboora
Department of Biology, Science Faculty, Alzahra University, Tehran, Iran