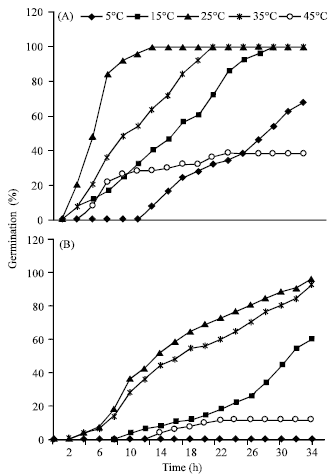
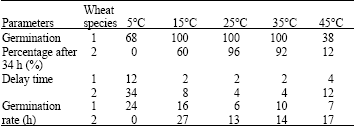
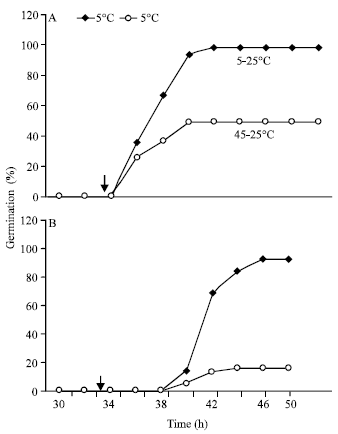
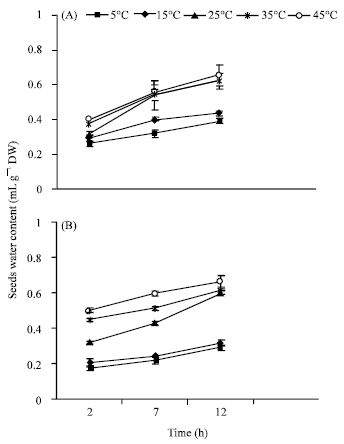
Research Article
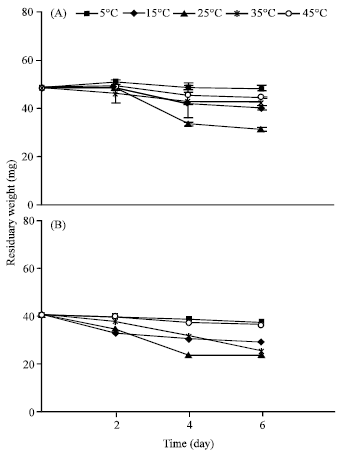
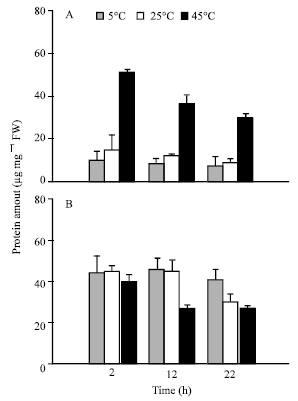
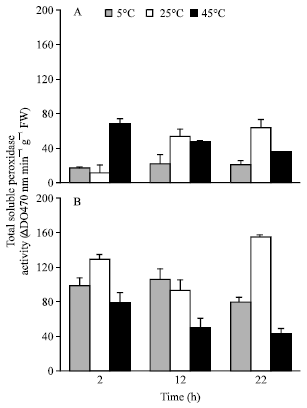
Sensitivity of Two Wheat Species’s Seeds (Triticum durum, Variety Karim and Triticum aestivum, Variety Salambô) to Heat Constraint During Germination
Departement des Sciences Biologiques, Laboratoire de Physiologie et Biotechnologie Vegetales Faculte des Sciences de Tunis, 1060, Tunisia
Saida Ammar
Departement des Sciences Biologiques, Laboratoire de Physiologie et Biotechnologie Vegetales Faculte des Sciences de Tunis, 1060, Tunisia
Najoua Jbir
Departement des Sciences Biologiques, Laboratoire de Physiologie et Biotechnologie Vegetales Faculte des Sciences de Tunis, 1060, Tunisia
Sadok Bouzid
Departement des Sciences Biologiques, Laboratoire de Physiologie et Biotechnologie Vegetales Faculte des Sciences de Tunis, 1060, Tunisia