Research Article
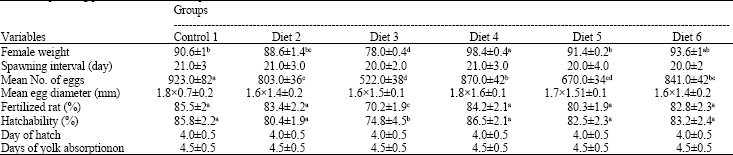
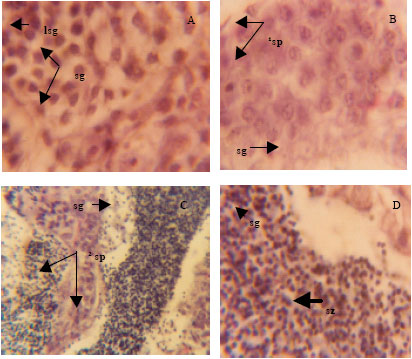
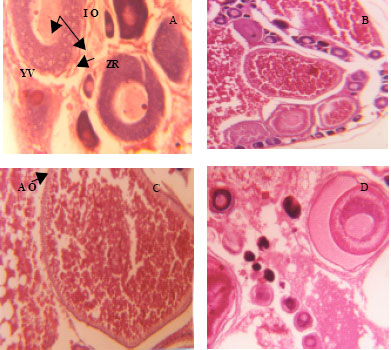
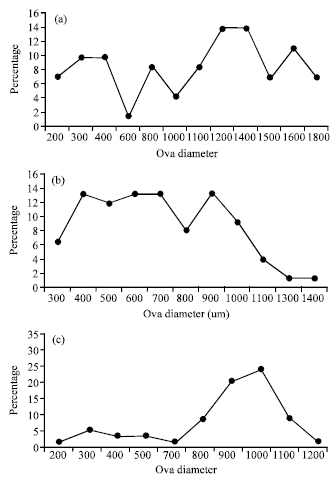
Effect of Varying Dietary Protein Sources on Growth and Spawning Performance and Gonad Maturation of Red Tilapia Reard in Sea Water
National Institute of Oceanography and Fisheries, Alexandria, Egypt
F. A. Raky
National Institute of Oceanography and Fisheries of Kanater el Khairia, Egypt










S.K.Prabhakar Reply
This paper is unique and usefull for research work.