Research Article
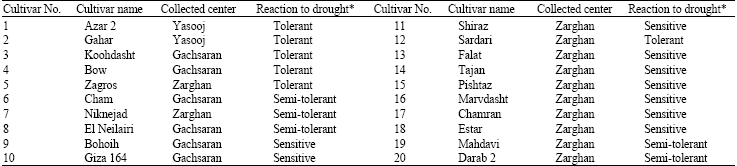
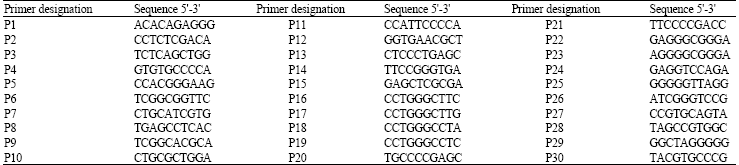
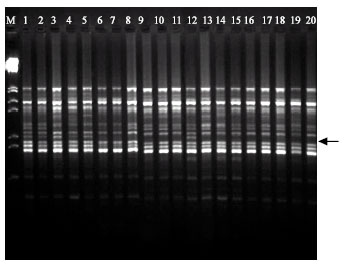
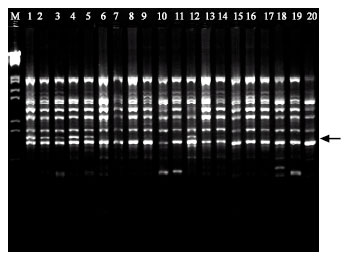
RAPD Markers Associated with Drought Tolerance in Bread Wheat (Triticum aestivum L.)
Department of Crop Production and Plant Breeding, Shiraz University, Shiraz, Islamic Republic of Iran
E. Tavakol
Department of Crop Production and Plant Breeding, Shiraz University, Shiraz, Islamic Republic of Iran