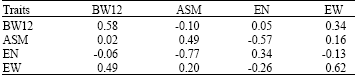
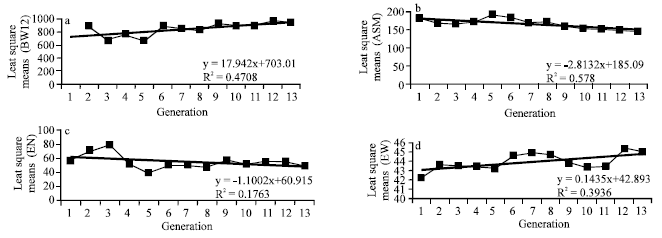
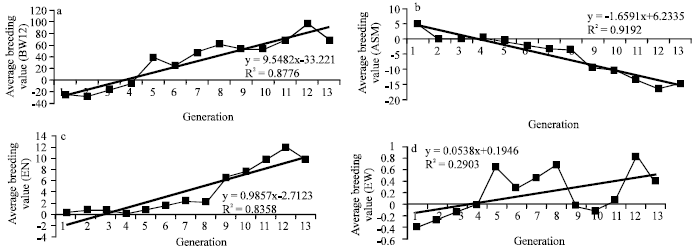
Research Article
Genetic Trend in Economic Traits in Iranian Native Fowl
Department of Animal Breeding and Genetics, Animal Science Research Institution of Iran (ASRI), Karaj, Islamic Republic of Iran
M. A. Kamali
Department of Animal Breeding and Genetics, Animal Science Research Institution of Iran (ASRI), Karaj, Islamic Republic of Iran