Research Article
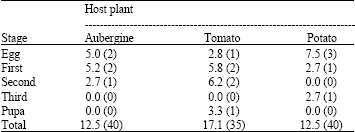
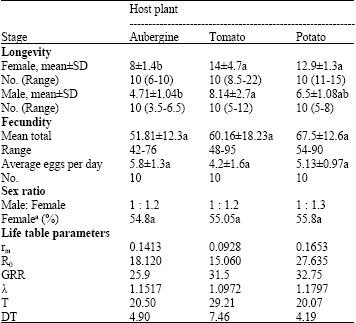
Some Biological Features of Cotton Whitefly, Bemisia tabaci (Homoptera: Aleyrodidae) on Various Host Plants
Department of Plant Protection, Faculty of Agriculture, Shahid Chamran University, Ahwaz, Iran
P. Shishehbor
Department of Plant Protection, Faculty of Agriculture, Shahid Chamran University, Ahwaz, Iran