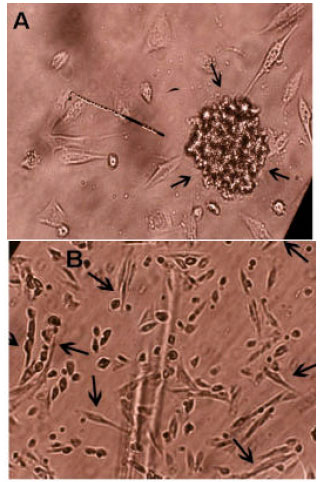
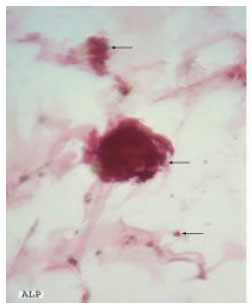
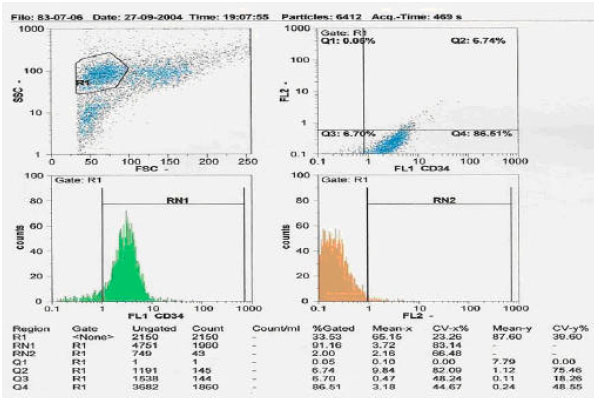
Research Article
Alkaline Phosphatase and CD34 Reaction of Deciduous Teeth Pulp Stem Cells
Razi Serum Research Institute, Karaj, Iran
Tahereh Foroutan
Department of Biology, Faculty of Basic Science, Tarbiat Moalem University, Tehran, Iran
Leila Jahangiri
School of Pharmacy, Mashbad University of Medical Sciences, Mashhad, Iran