Research Article
Effect of Thymoquinone on Malformations and Oxidative Stress-induced Diabetic Mice
Department of Medical Laboratory Sciences, College of Applied Medical Sciences, Al-JoufUniversity, P.O. Box 2014, Sakaka, Al-Jouf, Saudi Arabia
The co-existence of pregnancy and diabetes mellitus has been associated with congenital malformations, neonatal metabolic abnormalities, delayed development and fetal or maternal death (Yang et al., 2006; Galindo et al., 2006). Clinical and experimental evidences show the involvement of free radical-mediated oxidative processes in the pathogenesis of diabetic complications (Damasceno et al., 2002). It has been suggested that, supplementation of antioxidants during gestation period inhibits the genesis of free radicals which may decrease the rate of malformations, absorptions and abortions (El-Bassiouni et al., 2005; Padmanabham et al., 2006). Furthermore, it has been demonstrated that antioxidative therapy blocks diabetes-induced embryonic dysmorphogenesis in vivo and in vitro (Eriksson and Siman, 1996), although the mechanism for this protection remains to be elucidated. On the other hand, it is known that good perinatal outcomes can be achieved in diabetic pregnancies only by normalizing glucose values (Rosenn et al., 1994; Nasrat et al., 1996).
Thymoquinone is an active constituent of the volatile oil of the Nigella sativa L. has demonstrated strong antioxidant properties (Houghton et al., 1995). Nigella sativa L. family Ranunculaceae is widely used in traditional folk medicine in all Islamic countries. It has been shown to protect non-tumor tissues from chemotherapy-induced damage (Badary et al., 1997; Al-Shabanah et al., 1998) and suppresses expression of inducible nitric oxide syntheses in rat macrophages (El-Mahmoudy et al., 2002). Thymoquinone was reported to reduce the hepatic glucose production in diabetic hamsters (Fararh et al., 2005).
However, an antioxidant action has been reported to play an important role in the reproductive performance. Thymoquinone is well known as an antioxidant. The aim of this study is that to evaluate the effects of thymoquinone on reproductive performance and embryonic development in STZ-induced diabetic mice.
The present study is performed during 2004 at Quality Control Laboratory, Experimental Animal Care Center, College of Pharmacy, King Saud University, Riyadh, Kingdom of Saudi Arabia.
Thymoquinone (2-isopropyl-5-methyl-1,4-benzoquinone) (obtained from Sigma Co) was dissolved by initial addition of 0.05% dimethyl sulphoxide (DMSO), followed by the addition of normal saline. The solution was administrated at a dose of 10 mg kg -1 body weight once daily by gastric gauge for the whole gestational period (from day-1 of pregnancy to day-19).
Streptozotocin-induced diabetes: Streptozotocin (obtained from Sigma Co) was injected intraperitonially with a single dose of STZ (45 mg kg -1 body weight). After 24 h, blood samples were drawn from eye, by using a capillary tube and glucose concentrations (mg dL-1) were estimated by diagnostic kit available in the market (Randox laboratory, USA).
Animals and Housing: Virgin SWR female albino mice obtained from Experimental Animal Care Center, College of Pharmacy, King Saud University, Riyadh. Animals were maintained under controlled condition of temperature (24±1°C), humidity (50-55%) and light (12 h light/dark cycles) and were provided with Purina chow and water ad lib. The animals were divided in four groups for each parameters, 10 mice each, were used in each group, (I) control group (control animals were injected with an equal volume of the DMSO solution), (ii) animals were injected with THQ alone, (iii) animals were injected with STZ, (iv) animals were injected with STZ and THQ. Animals in groups (iii) and (iv) were injected with STZ before mating.
Induction and course of pregnancy: All groups of mice were introduced to a fertile stock of normal males. Vaginal plugs were checked every day for a period of five days, examined by eye, using forceps to open the vagina. The day vaginal plug was first seen, designated as day one of pregnancy (Gosden and Fowler, 1978). The pregnant female mice were sacrificed using carbon dioxide inhalation and maternal livers were removed and stored at -70°C for malondialdehyde and glutathione estimation. Fetuses and placenta were quickly removed from the uteri and weighed. Live, dead, total and resorptions fetuses were counted. Fetus morphology was examined according to body rotation, closure of the neural tube, appearance of heart, head, eyes, ears and pharyngeal arches. Fetuses with anomalies in one or more of these structures were denoted malformed.
Estimation of glutathione (GSH): Glutathione concentrations in liver of mice were estimated according to the method of Sedlak and Lindsay (1968). A cross sectional piece of the whole liver tissue was dissected and homogenized in ice-cold 0.02 M ethylene diamine tetra-acetic acid (EDTA). Aliquots of 0.5 mL of the tissue homogenates were mixed with 1.5 mL of 0.2 M Tris buffer, pH 8.2. Test tubes were closed with rubber caps and allowed to stand, with occasional shaking for 15 min before centrifuged at 3000 rpm at room temperature for 15 min. The absorbance of the clear supernatants was read using spectrophotometer at 412 nm, in 1 cm quarts cells. The concentrations of GSH were estimated using the standard curve.
Estimation of Malondialdehyde (MDA): The method described by Fong et al. (1973) was used with little modifications to estimate MDA concentrations in liver. Liver tissues were homogenized in Trichloro Acetic Acid (TCA) and the homogenates were suspended in thiobarbituric acid reagent (0.375 g thiobarbituric acid, 15 g trichloroacetic acid and 2.5 mL of concentrated hydrochloric acid in 100 mL distilled water) in test tubes and placed in a boiling water bath for 15 min. Tubes allowed to cool down at room temperature and centrifuged at 3000 rpm for 15 min. Optical density of the clear pink supernatants were read spectrophotometrically at 532 nm against reagent blank by using malondialdehyde bis (dimethyl acetal) as a standard.
Statistical analysis: The various parameters studied were subjected to statistical analysis with Student’s t-test with significance levels at p<0.05, 0.01 and 0.001. Results are expressed as mean values±SE.
Thymoquinone treatment during gestation period did not show any significant changes on total number of fetuses and had no effect on the weight of fetuses or placenta as compared to control. In STZ-induced diabetic mice, significant (p<0.001) reduction in the live fetuses (7.86±0.56) and increase (3.67±0.32, p<0.001) in fetus mortality compared to control group of mice (10.98±0.18 and 1.08±0.09) were observed (Table 1). Mean weight of fetuses (569.47±7.82) and placenta (62.36±3.47) were also significantly reduced, respectively (p<0.01) and (p<0.05) as compared to controls (623.84±11.81) and (76.36±3.94), respectively. However, the treatment with THQ during gestation period to diabetic mice, significantly reduced (p<0.01) (1.97±0.34) the number of dead fetuses and increased (p<0.05) the number of live fetuses (9.87±0.45) as compared to STZ-induced diabetic mice (3.67±0.32 and 7.86±0.56). The significant effect of THQ was seen on weight of the fetuses (606.81±8.72, p<0.05) compared to the diabetic mice (569.47±7.82), while the weight of placenta (70.38±2.16) was not affected (Table 1).
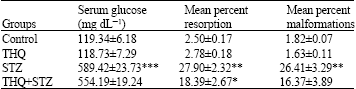
Mean percent resorptions were significantly (p<0.01) increased (27.90±2.32) in diabetic mice compared to controls (2.50±0.17). Thymoquinone treatment reduced (p<0.05) the percent resorptions in diabetic mice (18.39±2.67) as compared to diabetic mice (27.90±2.32, Table 2). Malformations were significantly high (p<0.01) in mice treated with STZ (26.41±3.29), but was reduced (16.37±3.89) and was not significant with THQ (Table 2).
| Table 1: | Mortality, fetal and placental weight on gestational day 19 |
 | |
*p<0.05, **p<0.01 and ***p<0.001 Student’s t-test. Groups THQ and STZ were compared with control and group THQ+STZ was compared with STZ group | |
| Table 2: | Effect of thymoquinone on diabetic embryopathy |
 | |
*p<0.05, **p<0.01 and ***p<0.001 Student’s t-test. Groups THQ and STZ were compared with control and group THQ+STZ was compared with STZ group | |
| Table 3: | Effect of thymoquinone on hepatic MDA and GSH concentrations |
 | |
*p<0.05, **p<0.01 and ***p<0.001 Student’s t-test. Groups THQ and STZ were compared with control and group THQ+STZ was compared with STZ group | |
Diabetic mice had higher glucose levels (589.42±23.73, p<0.001) compared to the mice in control group (119.34±6.18) (Table 2).
Liver MDA concentration was significantly (p<0.001) elevated in diabetic mothers as compared to normal mothers. Treatment with THQ significantly (p<0.01) reduced MDA as compared to the animals in diabetic group. In the mean time, glutathione level in mice liver was significantly reduced (p<0.01) by STZ injection as compared to controls; but significantly increased (p<0.05) by THQ treatments compared to the diabetic mice (Table 3).
Increased glucose metabolism by embryos resulting from maternal hyperglycemia disturbs a complex network of biochemical pathways, increases oxidative stress in embryos, generates Reactive Oxygen Species (ROS) and also depletes anti-oxidant defense. Oxidative stress has been implicated in diabetic embryopathy (Viana et al., 1996; Siman and Eriksson, 1997; Sivan et al., 1996; Wentzel and Eriksson, 1998). The present study examined the effects of THQ, extracted from Nigella sativa seeds, on hyperglycemia induced by STZ during pregnancy. In addition, glutathione and malondialdehyde were estimated. Malondialdehyde is generated by the breakdown of lipid hydroperoxides initiated by ROS (Harold et al., 2001).
It is well established that STZ functions by generating ROS (West, 2000). Streptozotocin is a selective pancreatic β-cell genotoxicant, when administered in a single high dose induces rapid onset of diabetes by generating DNA adducts, including N3-methyladenine and O (6)- methylguanine adducts and subsequently β-cell death by necrosis (Okomoto and Yamamoto, 1983; Cardinal et al., 2001; Burns and Gold, 2006). Damasceno et al. (2002) reported an increased incidence of skeletal and visceral malformations in fetuses from diabetic rats and attributed this to oxidative stress. This is in agreement with our results concerning the increase of resorption and malformations in mice treated with STZ alone.
Thymoquinone was shown to inhibit efficiently iron-dependent microsomal lipid peroxidation suggesting that it is a potent superoxide scavenger (Badary et al., 2003). It also acts as anti-cancer, immunomodulator and an anti-inflammatory (Salem, 2005; Gali-Muhtasib et al., 2006; El Gazzar et al., 2006). The results of present investigation showed a decrease in the percentages of resorption and malformations in the group of mice treated with STZ and THQ. This suggests that, this could be due to normal glucose levels attained by the mothers because of THQ which is shown to lower glucose level mediated through a decrease in hepatic gluconeogenesis (Fararh et al., 2005) and thus create a normal environment for the continuation of pregnancy.
Reactive oxygen species generation in embryos under hyperglycemic conditions has not been directly demonstrated. Oxidative stress is implicated by indirect evidence, such as phospholipids peroxidation and depletion of antioxidant glutathione (Cederberg et al., 2001). Depletion of glutathione was demonstrated to cause defective development in rat embryos exposed to hyperglycemia (Sakami et al., 1999). The results of the present work indicated an increase in malondialdehyde and decrease in GSH concentrations.
This might suggest that hyperglycemia disturbs the biochemical pathways and restores when the glucose level normalized due to the treatment of STZ-induced diabetics with THQ. The results of this study suggest that antioxidant treatment of women with diabetes might be important to prevent congenital malformations.
I am grateful to the Research Center, College of Pharmacy, King Saud University for the financial support and facilities to carry out this study (Research project No. CPRC141).