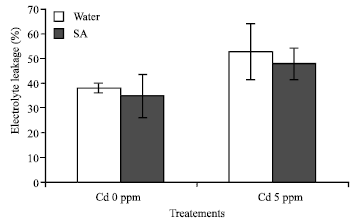
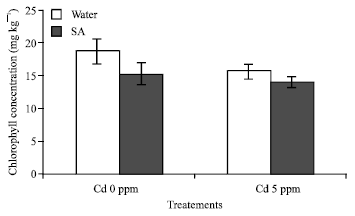
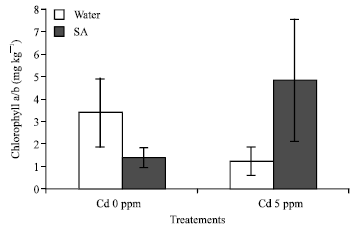
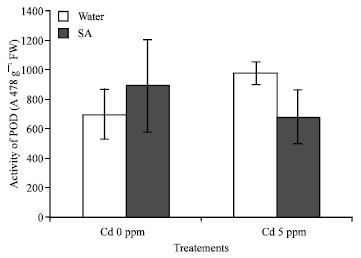
Research Article
Salicylic Acid Alleviates the Cadmium Toxicity in Chinese Cabbages (Brassica chinensis)
Department of Environmental Science, School of Resource and Environmental Science, Wuhan University, Wuhan 430079, China
Xiong Zhi-Ting
Department of Environmental Science, School of Resource and Environmental Science, Wuhan University, Wuhan 430079, China
Qiu Hai-Jie
Department of Environmental Science, School of Resource and Environmental Science, Wuhan University, Wuhan 430079, China