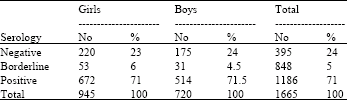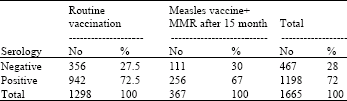Research Article
Prevalence of Human Herpes Virus-8 and Hepatitis B Virus among HIV Seropositive Pregnant Women Enrolled in the Mother-to-child HIV Transmission Prevention Program at Saint Camille Medical Centre in Burkina Faso
Laboratoire saint Camille de Ouagadougou, 01 BP 364 Ouagadougou 01, Burkina Faso, Italia
Damintoti Karou
Laboratoire saint Camille de Ouagadougou, 01 BP 364 Ouagadougou 01, Burkina Faso, Italia
Wendyame M.C. Nadembega
Laboratoire saint Camille de Ouagadougou, 01 BP 364 Ouagadougou 01, Burkina Faso, Italia
Aly Savadogo
Centre de Recherche en Sciences Biologiques, Alimentaires et Nutritionnelles (CRSBAN), UFR/SVT; Universite de Ouagadougou, 03 BP 7021 Ouagadougou 03 Burkina Faso, Italia
Ouermi Djeneba Salvatore Pignatelli
Laboratoire saint Camille de Ouagadougou, 01 BP 364 Ouagadougou 01, Burkina Faso, Italia
Virginio Pietra
Laboratoire saint Camille de Ouagadougou, 01 BP 364 Ouagadougou 01, Burkina Faso, Italia
Augustin Bere
Laboratoire saint Camille de Ouagadougou, 01 BP 364 Ouagadougou 01, Burkina Faso, Italia
Jacques Simpore
Universitcl di Roma Tor Vergata, Italia
Alfred S. Traore
Centre de Recherche en Sciences Biologiques, Alimentaires et Nutritionnelles (CRSBAN), UFR/SVT; Universite de Ouagadougou, 03 BP 7021 Ouagadougou 03 Burkina Faso, Italia