Research Article
Integration among the Different Aspects of the Phenotypic Plasticity in Aphis fabae, Black Bean Aphid
Department of Biology, Faculty of Science and Arts, Nigde University, 51100 Nigde, Turkey
INTRODUCTION
Recently it has been realized that phenotypic plasticity play an important role in speciation. Phenotypic plasticity (the capacity for individuals of the same genotype to express different phenotypes in different environments) had long been considered of lesser evolutionary importance because of its supposed lack of genetic basis and because phenotypic plasticity was often assumed to buffer the impact of natural selection and thus act to constrain speciation. As a consequence, there has been relatively less systematic study to analyze environmental effects on the phenotype and the evolutionary consequences of such plasticity. However, it has become increasingly clear that instead of restraining evolutionary change, phenotypic plasticity may actually foster it. Indeed, phenotypic plasticity may be a fundamental component of evolutionary change (Thompson, 1991; West-Eberhard, 2003; Görür, 2005).
Whilst there are plenty of studies which have determined plasticity in fitness components, morphological traits and host choice behavior, fewer have determined the relationships between these plasticities. Correlations among plasticities may indicate whether traits response to an environmental perturbation is systemic. The plasticity of one trait is related to the plasticity of another trait only if the two traits were genotypically and phenotypically correlated (Scheiner et al., 1991). For the life history traits, there should be selection for compensation, so that losses in fitness incurred by plastic changes in one trait are compensated for by increase in fitness due to plastic changes in another trait. This is main feature of integrated plastic responses. Various fitness components show integrated plastic responses as if 1) they share the same pool of resource and 2) they together respond to microenvironmental variation. If plasticities in different characters are correlated, selection causing differences among genotypes in the plasticity of any traits would create differences in the plasticities of other traits and the overall plastic response might be integrated (Scheiner et al., 1991; Stearns, 1992). In this context, relationships among plasticities of fitness components, morphological traits and preference-acceptance index (host choice) were examined.
Collection and maintenance of A. fabae genotypes: Thirteen Aphis fabae genotypes were used in this study. In order to increase genetic variability, the genotypes were sampled from several hosts and distinct locations. Collection sites were in the surrounds of Colchester, Essex, U.K., being at least 500 m apart from each other. Seven genotypes were collected on Euonymus europaeus (spindle), two from Vicia faba (broad bean), two from Chenopodium album (goosefoot) and one collected from Phaseolus sp. (French bean) and one from Tropaeolum majus (garden nasturtium).
Determining different aspects of phenotypic plasticities: In order to examine if environmental factors affect different aspects of phenotypic plasticities systemically, mean value and phenotypic plasticity of various features were measured. The intrinsic rate of natural increase (rm) and Relative Growth Rate (RGR) were calculated to determine the fitness of A. fabae genotypes on both host plants. Both rm and RGR are regarded as good indicators of aphid fitness. rm was calculated by the formula of Wyatt and White (1977).
rm= (0.738 * ln(Md))/d |
Where d: the development period from birth to beginning of first reproduction; Md: the number of nymphs born in the period from d to 2d days from birth. RGR was calculated based on the Scriber and Slansky’s (1981) equation.
RGR = ΔW/(Wx*d) |
Where = ΔW is the weight gained (Adult weight-Birth weight), Wx is the mean of adult weight plus birth weight divided by two and d is the development time. In order to evaluate fitness of each genotype, every newborn offspring weight and adult weight were measured as mg.
Calculation and analysis of morphological characters: Six morphological features for each genotype were measured under microscope using X100 magnification using slide-mounted individuals of all genotypes reared both on broad bean and nasturtium Adults from both host plants were used for morphological measurements. The six characters were: Ultimate rostral segment length (URS), the length of hind tarsal segment II (HT), siphunculi length (S), the distance between two siphunculi (SS), hind tibia segment length (TS) and the length of antennal segment III (AS).
Calculation and analysis of both preference and acceptance (Host choice): The host plant preference can be defined as the determination of the number of the species accumulate on the particular host plant (Singer, 2000). Preference experiments were carried out using plastic containers (26x18x10 cm) where the leaves of the potted plants tested were inserted through a hole made in the bottom of the boxes, which were then sealed with foam. The boxes were covered with transparent perspex sheets being all illuminated from above. For all clones 100 individuals were tested in trials using 20 individuals each time. Relative preference for broad bean was assessed by dividing the number of aphids preferring bean by the total number of aphids preferring broad bean and nasturtium.
The acceptance experiments were carried out in a controlled environment (insectary) reproducing the same environment conditions in which performance experiments were carried out. Acceptances of host plant were conducted in transparent acrylic boxes (35x35x55 cm). For each genotype at least three replicate groups were chosen. Ten adult females in each replicate were released onto a plant leaf by using a 00 grade paintbrush. These individuals were monitored regularly in order to record the number of aphids accepting each host plant. During the first 2 h, individuals were monitored every 30 min, then hourly up to 6 h and the final acceptance was recorded 24 h later from the beginning of the experiment. Aphids were monitored using a XLO magnifier glass to ensure they were feeding on the plant. After recording the final acceptance on one plant, these individuals were released on to another host plant and same procedure was followed.
The acceptance index (Ai) was calculated to summarize acceptance from 0 to 1. Acceptance index (Ai) is defined as:
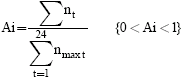 |
Where nt: No. of aphids feeding at time t, nmaxt: the number of aphids in the sample.
Assessment of phenotypic plasticities: Phenotypic plasticity was measured as the difference between the genotypic mean phenotypes across host plants for fitness. A two-way mixed model ANOVA with genotype (clones) and environments (hosts) as the factors was performed to evaluate phenotypic plasticity. Genotypes and hosts were considered random and fixed effects respectively (Zar, 1996).
The estimates of genetic correlation were calculated across host plants with Pearson’s product moment method, using genotypic means [(the rcm of Via, 1991)]. Genetic correlations within environments were estimated by the Pearson correlation coefficient.
RESULTS
Relationships among plasticity of fitness components: Phenotypic plasticities in birth weight, adult weight and development time were measured as the fitness components. Phenotypic plasticities in both the intrinsic rate of natural increase (rm) and Relative Growth Rate (RGR) were calculated as they are regarded as good indicators of aphid fitness. Results showed that there are generally positive relationships among plasticity of fitness components. Plasticity in fitness also mostly has positive correlations with plasticity in fitness components (Table 1, Fig. 1a and b).
| Table 1: | Relationships among plasticity of fitness components (Plasticity of Wb = birth weight, Wad = Adult weight, d = development time, F = relative fecundity, rm = fitness, RGR = relative growth rate) |
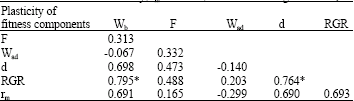 | |
| *p<0.05 after Bonferroni adjustment for multiple comparisons, Italic indicates almost significant value | |
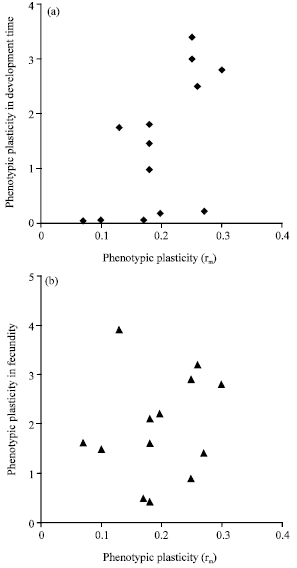 | |
| Fig. 1: | Relationships between plasticity in fitness and (a) phenotypic plasticity in developmental time, (b) Phenotypic plasticity in fecundity |
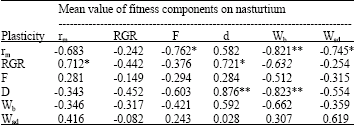
The relationships between plasticity of fitness components and mean value of these components on both hosts are different (Table 2 and 3). Plasticity in rm is generally positively correlated with rm on broad bean while it is negatively correlated on nasturtium (Fig. 2).
Plasticity in morphological characters: There are positive correlations among the level of plasticity of characters as morphological characters are closely positively correlated on both host plants.
| Table 2: | Correlations between plasticity of fitness components and genotypic mean value of these components on broad bean (Wb = birth weight, Wad = Adult weight, d = development time, F = relative fecundity, rm = fitness, RGR = relative growth rate) |
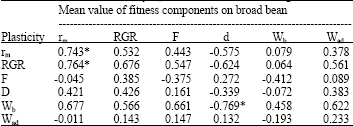 | |
| *p<0.05, **p<0.01 after Bonferroni adjustment for multiple comparisons, Italics indicate almost significant values | |
| Table 3: | Correlations between plasticity of fitness components and genotypic mean value of these components on nasturtium (Wb = birth weight, Wad = Adult weight, d = development time, F = relative fecundity, rm = fitness, RGR = relative growth rate) |
 | |
| *p<0.05, **p<0.01 after Bonferroni adjustment for multiple comparisons, Italics indicate almost significant values | |
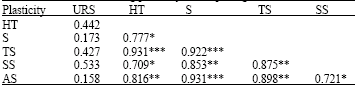
| Table 4: | Correlations among plasticity of morphological characters |
 | |
| *p<0.05, **p<0.0l, ***p<0.00l, after Bonferroni adjustment for multiple comparisons | |
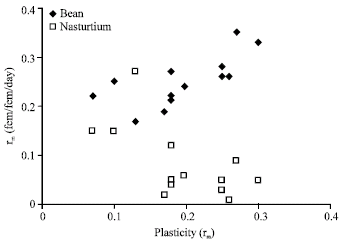 | |
| Fig. 2: | Relationships between plasticity in fitness and fitness measurement on both host plants |
Plasticity of ultimate rostral segment is generally non-significantly correlated with plasticity of other morphological characters (Table 4).
| Table 5: | Relationships between plasticity and genotypic mean of each morphological character on both hosts |
 | |
| *p<0.05 after Bonferroni adjustment for multiple comparisons | |
| Table 6: | Relationships between plasticity in host choice and plasticity in fitness and plasticity in morphological characters |
 | |
Plasticity in morphological characters are generally positively correlated with genotypic mean of these characters on broad bean, but they are negatively correlated on nasturtium.
The only exception is plasticity in ultimate rostral segment, which is highly positively correlated with genotypic mean of this character on nasturtium but not correlated on broad bean (Table 5).
Relationships between plasticity of fitness and plasticity of morphology: Plasticity in rm is generally weakly correlated with plasticity in morphological characters except plasticity in ultimate rostral segment (URS). Plasticity in URS is negatively genetically correlated with plasticity in rm (rcm= -0.572, non significant).
Relationships between plasticity in host choice and both plasticity in fitness and plasticity in morphology: Plasticity in rm is generally weakly related with plasticity in acceptance of both host plant. Plasticity in acceptance for broad bean reared genotypes is non-significantly positively correlated with plasticity in fitness but plasticity of nasturtium reared genotypes is non-significantly negatively correlated. Plasticity in acceptance for bean reared individuals is generally weakly positively correlated with plasticity in all morphological characters while plasticity of nasturtium reared genotypes is generally weakly negatively correlated (Table 6).
DISCUSSION
There are generally positive correlations among plasticity of fitness components. As it was shown on the Table 1 and Fig. 1a and b, plasticity in fitness also is positively correlated with other fitness components. There are also certain host plant effects on the plasticity in fitness and fitness measurement on nasturtium and broad bean as drawn from the Fig. 2. These findings showed that two host plants acted differently on relationships between both fitness in plasticity and correlations between plasticity and fitness measurements.
Relationships between plasticity and the average value of measured traits indicate that individuals with higher phenotypic plasticity display lower fitness on nasturtium in contrast to broad bean. In terms of morphology, individuals, that display higher phenotypic plasticity tend to show larger ultimate rostrum segment size but are smaller for other morphological characters on nasturtium in contrast on broad bean.
Table 4 shows close relationships among plasticity of morphological characters, which indicates systemic affects of host plants on plasticities in morphological features. There is only one exception, plasticity in Ultimate Rostral Segment (URS) is generally weakly correlated with plasticity of other morphological characters, indicating independent regulation of this trait.
There are not clear pattern to clarify the relationships between phenotypic plasticity in fitness components and morphological characters. These correlations indicate that both fitness components and morphological characters are not affected in a similar way from host plant conditions as correlations among plasticities indicate a systemic environmental effect on phenotypic plasticity (Scheiner et al., 1991; Stearns, 1992). Plasticity in fitness is weakly correlated with plasticity in morphological characters, except for plasticity in ultimate rostral segment. Although ultimate rostral segment has a significant positive relationship with fitness on nasturtium, plasticity in fitness is highly negatively correlated with plasticity in URS. The larger length on nasturtium, different relationships with other morphological characters and significant positive correlations with fitness on nasturtium strongly suggests that there is an adaptive value for the plasticity in ultimate rostral segment. The negative relationship between plasticity in ultimate rostral segment (URS) and fitness further emphasizes the adaptiye aspect of URS as fitness is very low on nasturtium.
There are also weak correlations between plasticity in host choice and plasticity in both fitness and morphology. These relationships indicate that plasticity of these features is affected differently by host plant conditions.
It can be concluded that although A. fabae genotypes showed higher amount of phenotypic plasticity in different aspects of phenotypic plasticities, there were not clear pattern to explain integration among them. The environmental effects for plasticity are thus not systemic for both this wide suite of traits and genetic effects may contribute to such differences.
I would like to thank to Cecilia Lomonaco de Paula and Aulay Mackenzie for their suggestions and helps during study.