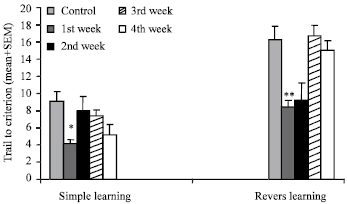
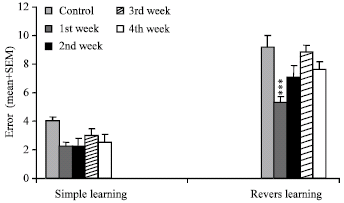
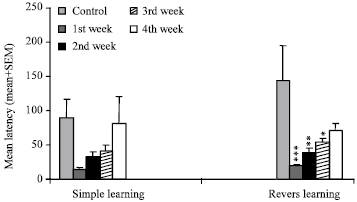
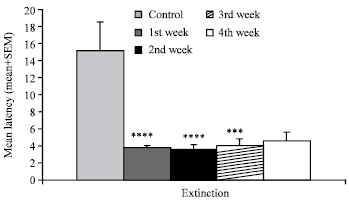
Research Article
The Effect of Palmitic Acid on Spatial Learning and Extinction in Adult Male Rat
Department of Biology,
Ahmad Ali Moazedi
Department of Biology,
Rahim Chinipardaz
Department of Statistics and Mathematics, Shahid Chamran University, Ahwaz, Iran