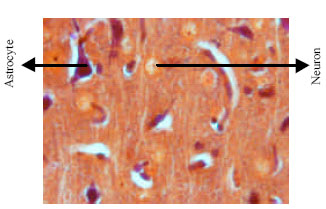
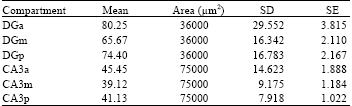
Research Article
The Similarity of Astrocytes Number in Dentate Gyrus and CA3 Subfield of Rats Hippocampus
Department of Anatomy, Gorgan University of Medical Sciences, Gorgan, Iran
Y. Sadeghi
Cellular and Molecular Research Center, Shahid Beheshti University of Medical Sciences, Tehran, Iran
A. Hosseini
Cellular and Molecular Research Center, Shahid Beheshti University of Medical Sciences, Tehran, Iran
N. Naghdi
Department of Physiology, Institute of Pastour, Tehran, Iran