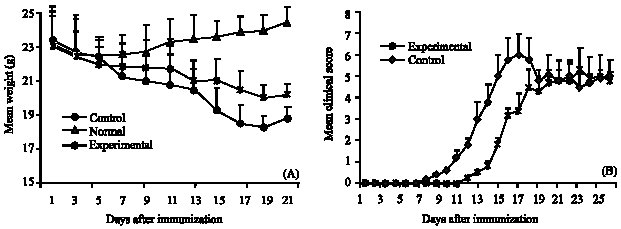
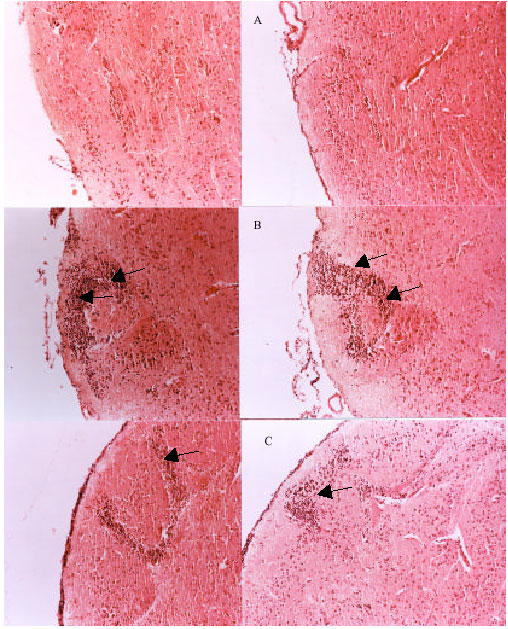
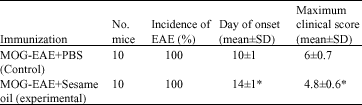
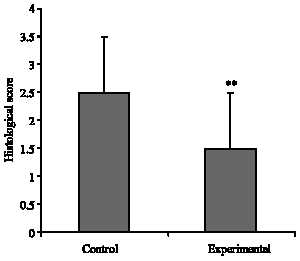
Research Article
Effect of Sesame Oil on the Inhibition of Experimental Autoimmune Encephalomyelitis in C57BL/6 Mice
Department of Histology, School of Medicine, Arak University of Medical Sciences, Arak, Iran
A. Ghazavi
Department of Histology, School of Medicine, Arak University of Medical Sciences, Arak, Iran
H. Salehi
Department of Histology, School of Medicine, Arak University of Medical Sciences, Arak, Iran
M. A. Payani
Department of Histology, School of Medicine, Arak University of Medical Sciences, Arak, Iran
M. R. Khazaei
Department of Histology, School of Medicine, Arak University of Medical Sciences, Arak, Iran