Research Article
Vibriosis as an Epizootic Disease of Rainbow Trout (Onchorynchus mykiss) in Turkey
Department of Fish Disease, Fisheries Faculty, Ege University, 35440 Urla, Izmir, Turkey
Vibriosis is one of the most prevalent fish diseases caused by bacteria belonging to the genus Vibrio. Vibriosis caused by Vibrio anguillarum has been particularly devastating in the marine culture of salmonid fish. The causative agent, V. anguillarum, was first described in 1909 by Bergman as the aetiological agent of the red pest of eels in the Baltic Sea. Vibriosis was not reported in North America until 1953.
When V. anguillarum was isolated from chum salmon (Oncorhynchus keta). Outbreaks affecting close to 50 species of fresh and salt water fishes have been reported in several countries in the Pasific, as well as the Atlantic coasts (Anderson and Conroy, 1970; Strout et al., 1978; Winton et al., 1983; Tolmasky et al., 1985). In Japan, vibriosis affects freshwater fish, especially ayu caused by V. anguillarum serotype J-O-1 in Lake Biva. In July 1990 the same disease killed more than 1,6 million feral ayu V. anguillarum has been demonstrated that the two serogroups, while 03 has been frequently isolated from diseased eels (Anguilla anguilla) (Larsen et al., 1994).
V. anguillarum was serotyped from O1 to O16 by Grisez and Ollevier (1995). Later, V. anguillarum strains, which were isolated from various fish species in all over the world, were reported in 23 O. serogroups (Pedersen et al., 1999).
V. anguillarum, also cause disease in marine fish aquaculture in Turkey (Cagirgan, 1993; Tanrikul et al., 2005) has been known for along time, but has not been previously reportedfrom rainbow trout (Oncorhynchus mykiss) in freshwater farming now causes many problems. And the infection spreads rapidly in rainbow trout farms.
The objective of this study was to determined, diagnosis confirmed serological and biochemical characteristics of V. anguillarum species. In addition, antibiogram results of isolated bacteria from fish farms are given. The strains included in this study were also selected based on diversity of geographical origin and in biochemical reactions that resulted from the preliminary identification.
Fish: Fish samples were made on juvenile and adult rainbow trout (Onchorynchus mykiss), ranging from 20 to 300 g of body weight from 8 different fish farm in Southern Aegean Region of Turkey. Recently died or moribund fish from 12 disease cases involving rainbow trout, were brought to Ege University, Fisheries Faculty, Department of Fish Disease’s Laboratory for disease diagnosis. Samples came from 8 fish farms, 6 land-based and 2 cage farms, during a 3 year period (2001-2004). The postmortem examinations of external and internal disease signs were performed.
Bacterial strains:
Isolation and selection: Kidney samples of 10 fish were inoculated onto Tryptic Soy Agar (TSA) (Oxoid) and Brain-Heart Infusion agar (BHA) (Oxoid), incubated at 21°C and examined daily for 3 days. Representative colonies on TCBS (Oxoid) were selected for purity. Those presumptively diagnosed as Vibrio sp. selected for further identification.
Preservation: Stock cultures were maintained at 4°C or at room temperature on TSA (Oxoid) and reinoculated every 1 or 2 months. All long-term storage of bacterial strains, were kept at -30°C on tryptic soy broth (Merc) supplemented with 10% glycerol.
Biochemical and physiological tests presumptive identification: Colony morphology was observed on TSA (Oxoid) and BHA (Oxoid) after 2 days of incubation at 21°C. Gram staining and oxidase test were carried out according to standard procedures. Motility was determined by the hanging drop method. Oxidative and fermentative degradation of glucose and gas production were tested on MOF medium (Difco). Sensitivity to the vibriostatic agent was tested with discs (Oxoid) on TSA (Oxoid).
Further identification: Young cultures were used for the biochemical tests and incubated at 21°C. TSA was used as a base in all test media except for growth tests at different salt concentration. The salt requirement was tested using peptone water. Biochemical tests carried out in API 20E systems (BioMerieux S.A). The incubation was 72 h at 21°C.
Challenge tests : PBS was used for all suspensions and dilutions of bacteria in the challenge tests and for the viable counts. A bacterial suspension in 1 mL PBS was inoculated onto two TSA with a pasteur pipette, excess liquid removed and the plates dried. The growth was harvested with 3 mL of PBS after 19 h incubation at 21°C. Serial 10 fold dilutions were prepared and kept on ice and the fish challenged with 1 h.
Rainbow trout parr of ≈50 g from a smolt-producing facility with no known history of disease were transferred to the experimental facility 7 days before challenge. The fish were kept in freshwater 15°C.
The experiments using 1 no isolate were terminated and an attempt to determine LD50 was made using 1 No. isolate only. Thirty fish were inoculated with dilution of bacteria and 30 fish with PBS for control. The fish were injected intraperitoneally with 0.1 mL, such that the dose per fish was 1.6x106 CFU. The fish were observed daily during the experiment, dead samples were taken aseptically from.
Preparation of antisera and slide agglutination test: Slide agglutination test were carried out with all isolated strains using raised rabbit serum against V. anguillarum O1 (ATTC 43305) according to Toranzo et al. (1987).
Bacterial cells were grown on BHA plates at 24-48 h 25°C and several loopfuls of the cells were resuspenced in PBS to obtain a concentration of a Mc Farland Standart No. 3. Those whole cell suspensions we used as antigens in the slide agglutination assays. A distict and immediately occurring agglutination was registered as positive and weak agglutination after 5 min was considered as negative test.
Antibiotic susceptibility testing: Drug resistance patterns of the isolates were determined by the disc diffusion method on Muller-Hilton agar (Difco), using the nine antibiotics (Oxoid and bioMerieux). Incubation on Muller-Hilton agar was carried out at 21°C at 24 h.
Clinical findings: The disease occurred in land-based farms in the summer and water temperature was recorded as 15°C and cage farms in the autumn when water temperatures decreased to 13°C. The diseased fish showed erratic swimming, dark discoloration and exophthalmia; the mortality varied from 20 to 30% in the farms.
The external signs were haemorrhage in the anal and pectoral fins and on the abdomen and there were bilateral exophthalmia with haemorrhage in the eyes. In various parts of the body in diseased fish, especially on the flanks skin lesions as ulcers were detected. Diffuse or petechial haemorrhage on the muscle was also noted.
In internal organs, a diffuse or petechial haemorrhage in liver, pyloric caeca, peritoneal or subperitoneal membranes, perivisceral fat and lower intestines were frequent internal signs.
Challenge tests: In all, 18 out of 30 fish infected with the agent died and none of control fish died. All fish except for control group that were infected with 1.6x106 CFU died after 9 days. Most of fish that died had haemorrhage and necrosis in the muscle at the injection site and petechial haemorrhage in the liver, pyloric caeca and the perivisceral fat. Bacteria were isolated from 18 fish samples that died during the experiment. No bacteria were isolated from the surviving fish.
Biochemical, physiological and serological tests: The morphologic and biochemical properties of isolated V. anguillarum strains are presented in Table 1. Microbiological analysis of internal organs rendered in all cases the growth in pure culture of motile Gram (-) bacteria. The isolates were found to be fermentative, oxidase and catalase positive, positive for dihydrolase arginine, negative for decarboxylation of lysine and ornithine, positive for hydrolisis gelatine and sensitivity to O/129. These characteristics allowed their presumptive identification as V. anguillarum. In a slide agglutination test with anti V. anguillarum O1 (ATCC43305) serum, showed that all strains were positive.
| Table 1: | The morphologic and biochemical properties of isolated V. anguillarum |
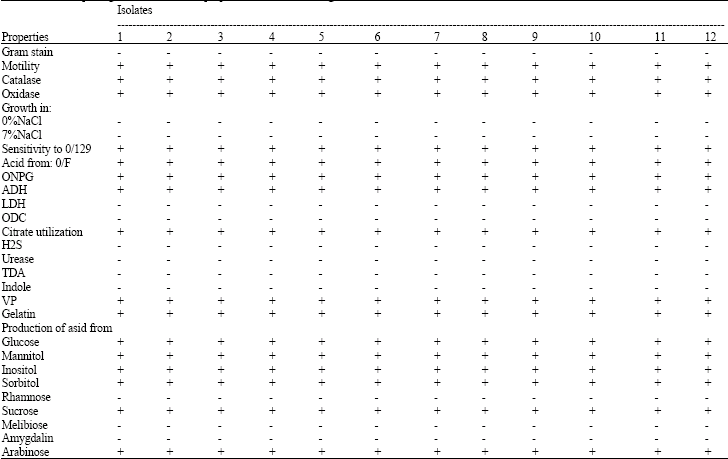 | |
+: Positive, -: Negative | |
| Table 2: | Antibiotic susceptibility profile of the isolates of V. anguillarum |
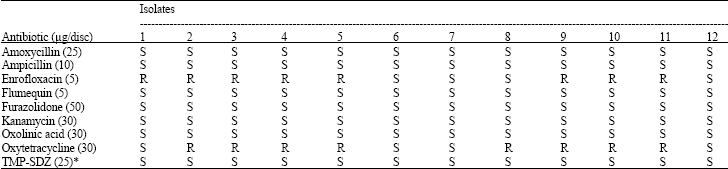 | |
S*Trimethoprim-Sulphadiazine(1/5), S: Sensitive, R: Resistant | |
Antimicrobial susceptibility testing: The antibiotic susceptibilities of the isolates of V. anguillarum from rainbow trout were assessed and are given in Table 2. Infected fish were treated with tribressen (Trimethoprim- Sulphadiazine 1/5) added to dry pellet feed (50 mg kg-1 fish per day) for 10 consecutive days and mortality was completely controlled.
The first report of vibrio disease occurring in fish raised in facilities in salt water or in fresh water was given by Rucker in 1954. However, that report dealt only with vibrio disease among Oncorhynchus gorbuscha, Oncorhynchus keta and Oncorhynchus tschawytscha raised in facilities along the Washington coast, there was no discussion of the sings or the pathogenic bacteria (Egusa, 1992). Reports on vibrio disease among salmonids have been increasing with their increased salt-water and fresh-water cultivation in Europe and the United States. Vibrosis reported that it diagnosed on Oncorhynchus tschawytscha in brackish water regions (Cisar and Fryer, 1969) and Oncorhynchus gorbuscha and Oncorhynchus keta in salt-water facilities (Holt, 1970; McCarthy et al., 1974; Ghittino et al., 1975; Egidius and Anderson, 1977). Muroga (1975) isolated V. anguillarum from Salmo gairdnerii irideus raised in salt water in Japan. Vibriosis occurs frequently among ayu (Plecoglossus altivelis) fingerlings raised after capture. Muroga and Egusa (1967) have examined the bacteria isolated from diseased ayu raised in Lake Hamana and they have reported the identification of V. anguillarum. Eight serotypes of V. anguillarum are usually isolated in Japan, (Ezura et al., 1980). Almost all the strains of V. anguillarum isolated from ayu killed by vibriosis in freshwater were serotype J-O-1 and those in seawater were serotype J-O-3. One of the characteristics of vibriosis in Lake Biwa is that mass killing of ayu have occurred not only in cultured populations but also in feral population (Eguchi et al., 2000).
Vibriosis appears in mariculture facilities in Turkey, V. anguillarum O1, V. ordalii were isolated from sea bass and V. anguillarum O1, V. alginolyticus was isolated from sea bream (Cagirgan, 1993; Tanrikul et al., 2005). Yersinia ruckeri (Cagirgan and Yurekliturk, 1992, 1998; Tanrikul, 1995) Lactococcus sp. (Cagirgan and Tanrikul, 1997) and Aeromonas salmonicida (Kirkan et al., 2003) were isolated from rainbow trout in freshwater farms. Kirkan et al. (2003) reported that the Aeromonas salmonicida was isolated from 265 rainbow trouts, which showed clinical sings from some rainbow trout farms and hatcheries in Turkey.
Vibriosis has appeared for the recently in rainbow trout farms in Turkish. Indeed, the disease in the intensive farming regions is spreading rapidly from farm to farm. On the contrary, vibrio species from salmonids were not isolated in natural streams in Japan until now (Eguchi et al., 2000).
There was biochemical variability existing among V. anguillarum O1 strains, which were isolated from sea bass. (Tanrikul et al., 2005). But from the O1 strains biochemical properties point of view, isolated V. anguillarum from different localities and different times in rainbow trouts did not show differences. Except fermentation of sugars, 3 isolates among the V. anguillarum O1 strains isolated from sea bass by (Tanrikul et al., 2005) showed similarities with rainbow trout isolates from the other biochemical properties. It is thought that feed with offal, which was obtained from marine fish, has played a major role in the disease contamination in rainbow trout farms. Because, vibriosis has first appeared in some farms, where is the fingerlings fed with this type of food in hatcheries. After, contamination spreaded to the cage culture lakes by transportation. Now, the disease often appears in both fish ponds and fish cages.
In antimicrobial susceptibility tests made from isolates to become isolated from vibriosis, were found to be sensitivity of bacteria to amoxycillin, ampycillin, flumequin, furazolidone, kanamycin, oxalinic acid and sulphonamides. Some of the isolates developed a resistance to enrofloxacin ve oxytetracycline. Because of the widespread enrofloxacin uses against the streptococcosis in rainbow trout farms in Turkey, V. anguillarum strains sensitivity also decreased against the chemotherapotic (Takahashi et al., 1976) recommended the sulphonamids in vibriosis treatment, (while Endo et al., 1975) proposed the oxolinic acid. Although V. anguillarum strains were also sensitive against the sulphonamids. But it was not applied in Turkey, so that, oxytetracycline was preffered. The resistances of bacteria were gradually raised against the antibiotic by applying oxitetracycline.
Aeromonas salmonicida was not isolated in this research. However (Kirkan et al., 2003) were isolated to Aeromonas salmonicida in the same area. So, it may be probably erroneously identification, because of the similarity of clinical symptoms of both diseases. In conclusion, in this study it is reported that Vibrio anguillarum O1 isolated from rainbow trout in freshwater farms of Turkey. Besides, results of antibiogram test were presented and identification of agent was supported with serologic tests.