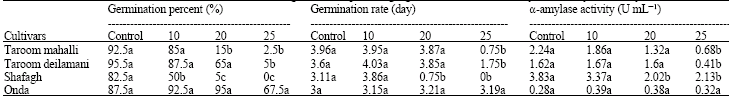
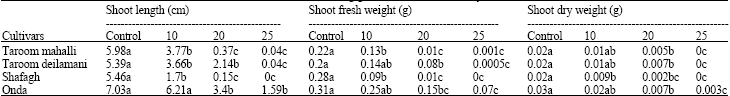
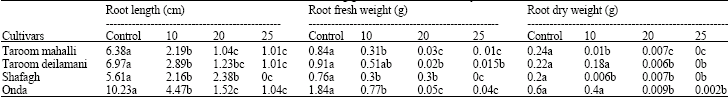
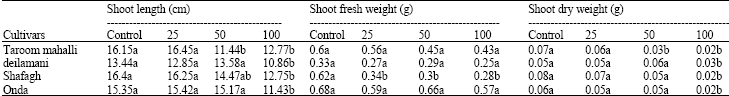
Research Article
Study of Growth Factors, α-amylase and Peroxidase Activity in Various Cultivars of Rice (Oryza sativa L.) Under Vanillic Acid Stress
Department of Biology, Faculty of Basic Science, University of Mazandaran, Babolsar, Iran
T. A. Aghajanzadeh
Department of Biology, Faculty of Basic Science, University of Mazandaran, Babolsar, Iran
B. Sadeghpour Gildeh
Department of Statistics, Faculty of Basic Science, University of Mazandaran, Babolsar, Iran