ABSTRACT
The antioxidant activity and the polyphenols and flavonoids contents of methanolic extracts of plant seeds of Canna indica, Hordium sativum and Capsicum annuum were investigated. These extracts were found to exhibit strong scavenging effects on 1, 1-Diphenyl-2-picryle-hydrazyl (DPPH) radicals and have significant amounts of flavonoids and polyphenols. The effects of these extracts on Phosphatidylcholine (PC) liposomes also were studied. It was found that the extractable materials have varying inhibitory effects on oxidation of PC liposomes and their aggregation. These effects were significant and illustrated that addition of these extractable materials to liposomes can enhance their stability by preventing liposome oxidation and aggregation.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/pjbs.2007.1643.1648
URL: https://scialert.net/abstract/?doi=pjbs.2007.1643.1648
INTRODUCTION
Liposome science and technology is one of the fastest growing scientific fields contributing to areas such as drug delivery, cosmetic, structure and function of biological membranes. This is due to several advantageous characteristics of liposomes such as ability to incorporate not only water soluble agents, specific targeting to the required site in the body and versatility in terms of fluidity, size charge and number of Lamellae (Mozafari, 2005).
Liposomes are colloidal structures formed by the self-assembly of amphophilic lipid molecules in solution (Tomohiro et al., 2002). Liposomal drug delivery systems have been widely researched for the reduction of drug toxicity and/or targeting of drug to specific cells (Mozafari, 2005; Pasquale et al., 2004). We believe that the efficiency of liposomes as a ‘vessel’ containing drugs is also important and there are many studies on dispersibility, micro viscosity, permeability of liposomes containing steroids, carboxyacyl phospholipids and water-soluble polymers (Shoko et al., 2002; Paraskevi et al., 2004; Yumiko et al., 2001) and on loaded antibiotic into liposomes (Ali et al., 2004) and their applications as models of biological membranes and as drug delivery vehicles for in vivo application (Lawrence et al., 1986).
Incorporation of flavonoids and α-tocopherol into lipid bilayer is some times the first step in the sequence of events induced by phenolic compounds, as change their fluidity, aggregation and oxidation (Sandra et al., 2003; Andrzej, 2006; Fatima et al., 2003).
Lipid auto oxidation is a free radical process which proceeds via a chain reaction including induction, propagation and termination steps. Antioxidant can interfere with the oxidation process by reacting with free radicals, chelating, catalytic metals and also by acting as oxygen scavengers (Concepcion et al., 1999); Wuguo et al., 1997; Kevin et al., 1999; Vitaly, 2003).
Plants contain a wide variety of free radical scavenging molecules such as flavonoids, anthocyanins, cartenoids, dietary glutathione, vitamins and endogenous metabolites and such natural products are rich in antioxidant activities. These plant-derived antioxidants have been shown to function as singlet and triplet oxygen quenchers, peroxide decomposers, enzyme inhibitors and synergists (Chang et al., 2002; Gow-Chin et al., 2002; David et al., 1998).
In recent years, plant-derived flavonoids have attracted increased attention because of their anticancer properties, which may be exerted through different biological effects. Among others, the flavonoids are known as potent antioxidants, cell growth inhibitors, multidrug resistance modulators, inhibitors of different types of kinases (Andrzej et al., 2002), anti-inflammatory, antithrombotic bactericidal and antiviral activity (Antonella et al., 1995; Chen et al., 1996), in addition, they possessing activity in both the hydrophilic and lipophilic systems (Arti et al., 1998; Zhiguo and William, 1999)
The aim of this study was to investigate the antioxidant activity of plant seed phenolic compounds and to illustrate their effects in soybean phosphatidylcholine liposomes (as model system) through preventing their lipid peroxidation and aggregation.
MATERIALS AND METHODS
Materials: L-Ascorbic acid and HCl (Surechem product LTD, England). Chloroform, Methanol (GCC Analyt/ Gainland Chemical Co. UK). Sodium carbonate (RIEDEL-Dehaen, Germany). Gallic acid, p-Dimethylaminocinnamaldehyde, Catechin and 1, 1-Diphenyl-2-picryl-hydrazyl (DPPH) (Sigma-Aldrich Chemie, Germany). Soybean Phosphatidylcholine (PC) (Phospholipid GMBH, Cologne, Germany). Tris-HCl (BDH Chemicals LTD. Poole, England). FeSO4.7H2O (Fluka/Purum. Ph. Eur. Switzerland). Trichloroacetic acid (Janseen, Chimica, Eur. Pharma; USP, Belgium). Thiobarbituric acid (Sigma-Aldrich Chemie Gmbh, Steinheim, Germany). Folin- Ciocalteau (KOCH-Light LTD, Haverhill-Suffolk, England.
Preparation of empty liposomes: Phosphatidylcholine (PC) (5 mg mL-1) in chloroform solution was dried to leave a thin film on the wall of round-bottom flask by removal of the organic solvent using a rotary evaporator (RV 05-ST Janke and Kunkel, IKA, Germany). The lipids film was hydrated with Tris -HCl buffer (100 mM, pH 7.5) followed by vigorous shaking. Unilamellar liposomes were obtained by ultrsonicate bath (Clifton, England) for 60 min, until the suspension became clear (Jizomoto et al., 1989; Kagan et al., 1990). For the experiments, 150 μL of methanolic extracts of Canna indica, Hordium sativum and Capsicum annum was added to PC chloroform solution, dried, evaporated and then sonicated as mentioned above.
Optical density: The optical densities of the liposome suspensions were measured at a visible light range from 350-650 nm (Trofimove and Nisnevich, 1990).
Liposome aggregation measurement: The absorbance of liposome dispersions measured at 400 nm was used as a qualitative measure of liposome aggregation. Measurement was performed with spectrophotometer (Milton Roy Company, Spectronic 1201, USA). For each sample, the absorbance was measured for 30 days after the addition of methanolic extract to liposome dispersions.
Liposome peroxidation assay (TBA method): The reaction mixture contained 500 μL of liposome solution, 500 μL Tris-HCl (100 mM, pH 7.5), 100 μL FeSO4.7H2O (4 mM). Liposome peroxidation was initiated by the addition of 100 μL of ascorbic acid (2 mM), incubated for 30 min at 37°C and terminated by the addition of trichloroacetic acid (5.5%). One milliliter of the mixture was added to 250 μL of Thiobarbituric Acid (TBA) in 50 mM NaOH, followed by heating for 10 min. The mixtures were centrifuged at 3500 rpm for 10 min and the absorbance of the supernatant was read at 532 nm with spectronic 1201 spectrophotometer (Chang et al., 2002).
Determination of total polyphenols: The amount of total phenolics in the extract was determined according to Folin-Ciocalteau method (Maria et al., 2006). The reaction mixture was consisted of 0.5 mL of the extract (1.5-15 mg mL-1); 0.5 mL of the Folin-Ciocalteau reagent, after a period of 3 min, 1 mL of saturated sodium carbonate solution was added. The 10 mL volumetric flasks were shaken and allowed to stand for 1 h. After 30 min, the absorbance was measured at 725 nm using spectrophotometer (Milton Roy Company, Spectronic 1201, USA). The concentration was calculated using gallic acid as standard and the results were expressed as milligram gallic acid equivalents per gram extract.
Determination of flavonoids: Dissolve about 100 mg of the seed extract in 25 mL of methanol and 1 mL of this solution was pipette into a test tube. A total of 5 mL of the chromogen reagent (1.00 g of 4-dimethylaminocinnamaldehyde dissolved in cooled mixture of 250 mL concentrated HCl and 750 mL methanol, complete to 1 L with methanol) was added to the extract solution and after 10 min the absorbance was measured at 640 nm using Spectronic 1201 spectrophotometer against a blank with water instead of extract solution. A calibration curve was prepared with (±) catechin and the results were expressed as milligram (±) catechin equivalents per gram extract (Matthaus, 2002).
Determination of antioxidant activity: Dissolve about 80 mg of the seed extract in 25 mL of methanol, sonicate for 20 min to dissolve it by ultrasonic bath (Clifton, England), then 2 mL of this solution was added to 3 mL of DPPH solution (50 mg/100 mL) and filled up with methanol to 25 mL. The mixture was shaken vigoursly and allowed to stand for 45 min in the dark. The decrease in absorbance was measured at 515 nm against a blank (without extract) with Spectronic 1201 spectrophotometer. The antioxidant activity was calculated using gallic acid as standard and the result were expressed as milligram gallic acid equivalents per gram extract (Matthaus, 2002).
Statistical analysis: All analysis were run in triplicate and averaged. Significant differences (p<0.05) were determined using Duncan’s range test.
RESULTS AND DISCUSSION
Liposome suspensions showed different optical densities (absorbance) at the spectral range 350-650 nm (Fig. 1). These difference may be due to incorporation of different methanolic extracted materials especially polyphenols and flavonoids of Cannna indica, Hordium sativum and Capsicum annuum into PC liposomes. This incorporation may induce changes in the structure of liposomal lipids and as a result changes occur in their properties.
The extracts tested in this study demonstrated vastly different inhibitory effects on rates of lipid peroxidation in liposomes. These small differences in antioxidant activity based on differences in the structure (hydroxyl group position) are probably a result of the enhanced planarity of the molecule (van Acker et al., 1996) and depends on the temperature and time of incubation. Figure 2 shows the effects of incorporation of different extracts to PC liposomes and how they reduce the liposome oxidation especially when incubated at 4°C (Fig. 2C), as compared to PC liposomes (control). Incubation of these liposomes at Room Temperature (RT) and 37°C enhanced lipid oxidation of liposomes especially the control one (Fig. 2A and B). It was noticed that there were slight differences between the incorporated extracts in preventing liposome oxidation; this may be due to the type of polyphenols and flavonoids in the extracts. For example, methanolic extract of plant seed Cannna indica has high effects in inhibiting the lipid oxidation compared to other methanolic extracts Hordium sativum and Capsicum annuum.
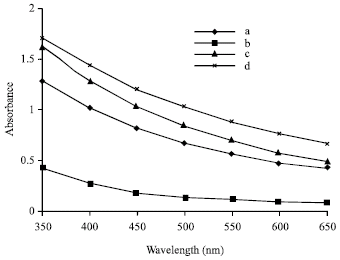 | |
| Fig. 1: | The optical densities of different types of liposomes at spectral range 350-650 nm. a: Phosphatidylcholine liposomes (control); Phosphatidylcholine liposomes with methanolic extract of b: Canna indica; c: Hordium sativum and d: Capsicum annuum |
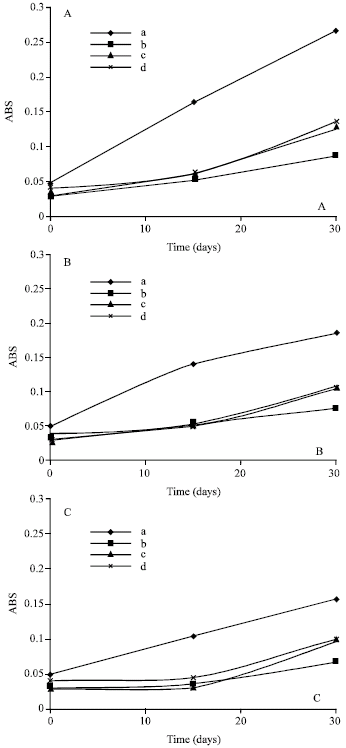 | |
| Fig. 2: | Effects of different extracts on liposome oxidation at different incubation temperatures for 30 days by TBA method measured at 532 nm. ( ♦ ): Phosphatidylcholine liposomes (control); Phosphatidylcholine liposomes with methanolic extract of: (■) Canna indica; (▲) Hordium sativum; (*) Capsicum annuum. A: Effects at 37°C; B: Effects at RT and C: Effects at 4°C |
These results were found to be related to the phenolic and flavonoids contents and to the antioxidant activity (Table 1).
| Table 1: | The concentration of flavonoids and polyphenols and DPPH antioxidant activity in methanolic extracts of plant seeds: Canna indica, Hordium sativum and Capsicum annuum |
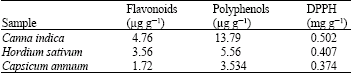 | |
| μg: microgram; mg: milligram; g: gram | |
Table 1 showed that methanolic extract of plant seed Cannna indica has the highest content of polyphenols and flavonoids and has a high antioxidant activity when compared to others.
The antioxidant activity of flavonoids tested was conspicuously related to the number and position of their aromatic hydroxyl groups. The effectiveness of an antioxidant is determined by many factors including its activation energy, rate constant, oxidation reduction potential, stability of the radical intermediate, thermal stability and hydrophobicity.
These results seem to demonstrate that some flavonoids alter the behaviour function of lipid bilayer, thus transferring from PC liposomes to the aqueous medium. Conversely, no change in membrane permeability appears to occur when liposomes are charged with some flavonoids. Similarly, Ikigai et al. (1993) reported that catechins can damage lipid bilayer.
This may be due to the flavonoids ability to interact with biomembranes, together with their redox properties, a fundamental requisite for the expression of their antioxidant activity (Saija et al., 1995).
According to the aggregation experiments, it was found that incorporation of these extracted materials into PC liposomes mimics their aggregation and there for enhance their stability by preventing the liposome oxidation. Control liposomes have higher percentage of aggregation when compared to the others especially at 37°C (Fig. 3A).
The time courses of absorbance of mixtures of PC liposomes with methanolic extracts are shown in the form of percentage (Fig. 3A-C). For all samples, their was increased percentage of aggregation in a time dependent manner, but with slight difference between samples containing liposomes with extracts and this may be due to the difference in the extracted materials especially those polyphenols and flavonoids.
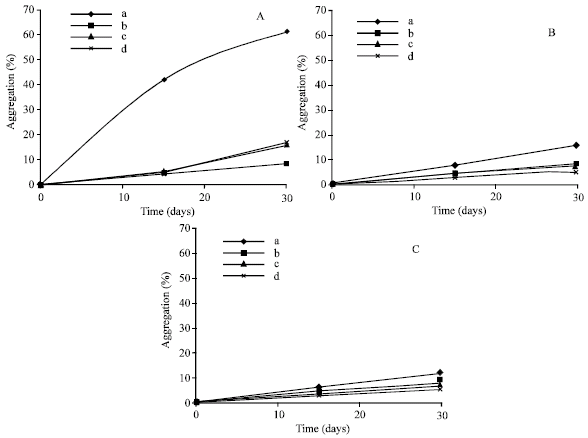 | |
| Fig. 3: | Effects of different extracts on liposome aggregation at different incubation temperatures for 30 days by measuring liposome suspension at 400 nm (in the form of percentage); (♦ ): Phosphatidylcholine liposomes (control); Phosphatidylcholine liposomes with methanolic extract of: ( ■) Canna indica; (▲ ) Hordium sativum; (*) Capsicum annuum, A: Effects at 37°C, B: Effects at RT and C: Effects at 4°C |
CONCLUSIONS
Our observations demonstrated the varying inhibitory effects of the three methanolic extracted materials specially flavonoids and polyphenols on oxidation of PC liposomes and their aggregation. The inhibitory characteristics of these materials depends on their contents of flavonoids and polyphenols, the structure of flavonoids specially their hydrophobicity/hydrophilicity, the total number and location of hydroxyl groups on aromatic rings, in addition to the antioxidant activity.
ACKNOWLEDGMENTS
I would like to thank Dr. Saleh Quran for his assistance in plant classification and Mr. Yassen Al-Quisi for his assistance in some experiments.
REFERENCES
- Sezer, A.D., A.L. Bas and J. Akbuga, 2004. Encapsulation of enrofloxacin in liposomes I: Preparation and in vitro characterization of LUV. J. Liposome Res., 14: 77-86.
Direct Link - Hendricha, A.B., R. Malona, A. Polaa, Y. Shiratakib, N. Motohashic and K. Michalaka, 2002. Differential interaction of sophora isoflavonoids with lipid bilayers. Eur. J. Pharm. Sci., 16: 201-208.
Direct Link - Antonella, S., B. Francesco T. Domenico, T. Antonio, M. Lucia, S. Patrizia and C. Francesco, 1995. Flavonoid-biomembrane interaction: A calorimetric study on dipalmitoylphosphatidycholine vesicles. Int. J. Pharm., 124: 1-8.
Direct Link - Arora, A., M.G. Nair and G.M. Strasburg, 1998. Structure-activity relationships for antioxidant activities of a series of flavonoids in a liposomal system. Free Radic. Biol. Med., 24: 1355-1363.
CrossRefDirect Link - Choi, C.W., S.C. Kim, S.S. Hwang, B.K. Choi and H.J. Ahn et al., 2002. Antioxidant activity and free radical scavenging capacity between Korean medicinal plants and flavonoids by assay-guided comparison. Plant Sci., 163: 1161-1168.
CrossRefDirect Link - Chen, Z.Y., P.T. Chan, K.Y. Ho, K.P. Fung and J. Wang, 1996. Antioxidant activity of natural flavonoids is governed by number and location of their aromatic hydroxyl groups. Chem. Phys. Lipids, 79: 157-163.
CrossRefDirect Link - Sanchez-Moreno, C., J.A. Larrauri and F. Saura-Calixto, 1999. Free radical scavenging capacity and inhibition of lipid oxidation of wines, grape juices and related polyphenolic constituents. Food Res. Int., 32: 407-412.
CrossRefDirect Link - Mantle, D., J.G. Anderton, G. Falkous, M. Barnes, P. Jones and E.K. Perry, 1998. Comparison of methods for determination of total antioxidant status: Application to analysis of medicinal plant essential oils. Comparative Biochem. Physiol. Part B, 121: 385-391.
Direct Link - Fatima, P.M., H.G. Michael and G. Paula, 2003. Activity and location of olive oil phenolic antioxidant in liposomes. Chem. Phys. Lipids, 124: 23-36.
Direct Link - Yen, G.C., P.D. Duh and H.L. Tsai, 2002. Antioxidant and pro-oxidant properties of ascorbic acid and gallic acid. Food Chem., 79: 307-313.
CrossRefDirect Link - Ikigai, H., T. Nakae, Y. Hara and T. Shimamura, 1993. Bactericidal catechins damage the lipid bilayer. Biochim. Biophys. Acta, 1147: 132-136.
PubMedDirect Link - Robards, K., P.D. Prenzeler, G. Tucker, P. Swatsitang and W. Glover, 1999. Phenolic compounds and their role in oxidative processes in fruits. Food Chem., 66: 401-436.
CrossRefDirect Link - Matthaus, B., 2002. Antioxidant activity of extracts obtained from residues of different oilseeds. J. Agric. Food Chem., 50: 3444-3452.
CrossRefDirect Link - Maria, A., K. Panagiotis, P. Vassilios, N. Andreana and B. Dimitrios, 2006. Radical scavenging activities of various extracts and fractions of sweet orange peel (Citrus sinesis). Food Chem., 94: 19-25.
Direct Link - Mozafari, R.M., 2005. Liposomes: An overview of manufacturing techniques. Cell. Mol. Biol. Lett., 10: 711-719.
PubMed - Kallinteri, P., D. Fatouros, P. Klepetsanis and S.G. Antimisiaris, 2004. Arsenic trioxide liposomes: Encapsulation efficiency and in vitro stability. J. Liposome Res., 14: 27-38.
Direct Link - Stano, P., S. Bufali, C. Pisano, F. Bucci and M. Barbarino et al., 2004. Novel camptothecin analogue (gimatecan)-containing liposomes prepared by the ethanol injection method. J. Liposome Res., 14: 87-109.
Direct Link - Antonella, S., B. Francesco T. Domenico, T. Antonio, M. Lucia, S. Patrizia and C. Francesco, 1995. Flavonoid-biomembrane interaction: A calorimetric study on dipalmitoylphosphatidycholine vesicles. Int. J. Pharm., 124: 1-8.
Direct Link - Verstraeten, S.V., C.L. Keen, H.H. Schmitz, C.G. Fraga and P.I. Oteiza, 2003. Flavan-3-ols and procyanidins protect liposomes against lipid oxidation and disruption of the bilayer structure. Free Radical Biol. Med., 34: 84-92.
CrossRefDirect Link - Imura, T., K. Otake, S. Hashimoto, T. Gotoh and M. Yuasa et al., 2003. Preparation and physicochemical properties of various soybean liposomes using supercritical reverse phase evaporation method. Colloids Surfaces B: Biointerfaces, 27: 133-140.
Direct Link - Van Acker, S., M.J. de Groot, D.J. van den Berg, M.N.J.L. Tromp, D.O.G. den Kelder, W.J.F. van der Vijgh and A. Bast, 1996. A quantum chemical explanation of the antioxidant activity of flavonoid. Chem. Res. Toxicol., 9: 1305-1312.
CrossRefDirect Link - Vitaly, R., 2003. Chain-breaking antioxidant activity of natural polyphenols as determined during the chain oxidation of methyl linoleate in Triton X-100 micelles. Arch. Biochem. Biophys., 414: 261-270.
Direct Link - Wuguo, D., F. Xing Wang and W. Jilan, 1997. Flavonoids function as antioxidants: By scavenging reactive oxygen species or by chelating iron. Radiat. Phys. Chem., 50: 271-276.
Direct Link - Yumiko, Y., T. Tadashi, I. Tomohiro, Y. Hitoshi, Y. Shoko, S. Hideki and A. Masahiko, 2001. Effect of adsorption of bovine serum albumin on liposomal membrane characteristics. Colloids Surfaces B: Biointerfaces, 20: 95-103.
Direct Link - Zhiguo, J. and J.B. William, 1999. Phenolics and lipid-soluble antioxidants in fruit cuticle of apples and their antioxidant activities in model systems. Postharvest Biol. Technol., 16: 107-118.
Direct Link








